The Technological Targeted Action Drug Discovery and Screening (ATC-DDS) coordinates projects and collaborations at the Institut Pasteur Paris campus on therapeutic development.
It raises awareness for DDS in basic research projects and stimulates intra-campus collaborations between Units, technological platforms and researchers with complementary expertise for innovative projects in the field of drug discovery. The ATC-DDS aims to strengthen collaborations within the Pasteur network and to increase national and international visibility in therapeutic development. It implements novel DDS technologies and anticipates potential future pandemics.
The ATC-DDS is led by Fabrice Agou, head of the Chemogenomic and Biological Screening Platform (PF-CCB) and Nienke Buddelmeijer, group leader of the Cell Shape and Pathogenicity group in the Biology and Genetics of the Bacterial Cell Wall Unit. They are advised by the ATC-DDS Scientific Advisory Board composed of Hélène Strick-Marchand, Nadia Naffakh, Iuliana Ene, Eric Prina, Ludovic Sauguet, Marcel Hollenstein, Olivier Sperandio and Christophe Zimmer. The SAB covers expertise in screening, in silico docking of molecules, adapted animal models to study human diseases, vaccine development strategies, structural biology and modeling, chemical approaches and artificial intelligence, on the scientific axes and concerted actions defined in the 2019-2023 strategic plan : Emerging Infectious Diseases, Antimicrobial Resistance, Brain Connectivity and Neurodegenerative Diseases, Cancer Initiative and Vaccinology and Immunotherapy initiative.
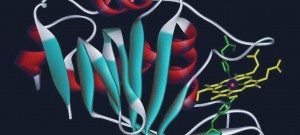
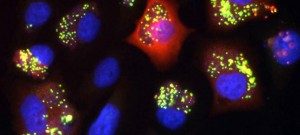
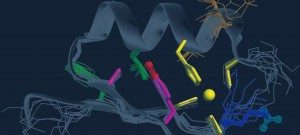
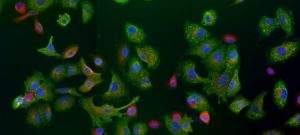
The PF-CCB platform has a large repertoire of state-of-the-art equipment to fulfill the needs of both phenotypic (High content screening HCS) and target-based (High-throughput screening HTS) screening projects on campus. To this aim, the platform has already developed biochemical, cellular and fluorescence microscopy screening tests and plans to extend its expertise in screening (i) by spatio-transcriptomic imaging on single cells, from organoids or patient tissues (Rebus Biosystems, Santa Clara, USA) and (ii) by high-throughput microfluidic interferometry (Creoptix, Switzerland).
Thanks to the ATC-DDS, the PF-CCB equipment was reinforced and the compound library collection at Institut Pasteur Paris is diverse and has been extended since 2021 with 7 new compound libraries, providing a total of 27 774 new compounds to the community. Four short-term research projects were financed under the ATC-DDS trainee call 2021 that all focused on the development or improvement of biological assays for future screening for antibacterial and antiviral molecules. Our IP expertise in DDS was shared both in France and in Europe through 7 yearly international lectures at Radboud University (Netherlands), and a yearly masterclass & accelerated learning series at Sup’Biotech (France) — all coordinated by Fabrice Agou and the PF-CCB. Finally, 2 full days DDS symposia and 2 seminars with international invited guests with expertise un the DDS field were organized on the campus for Pasteur researchers and external participants.