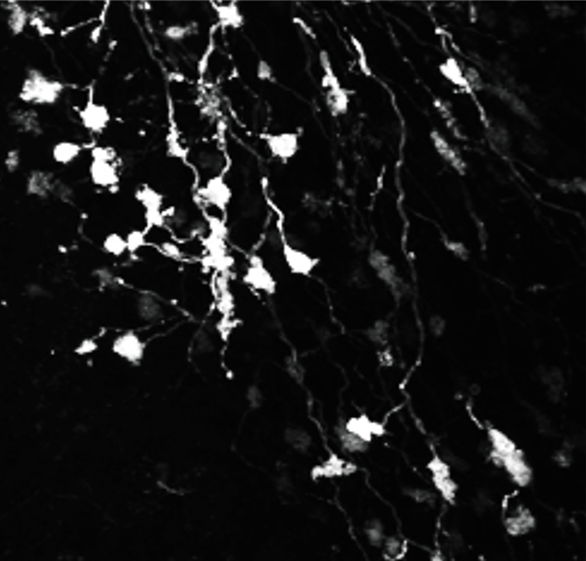
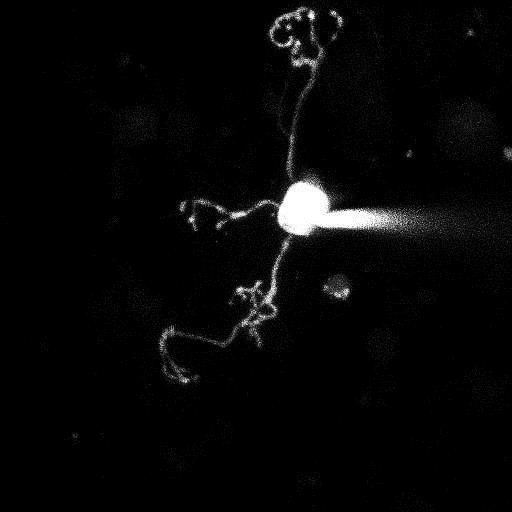
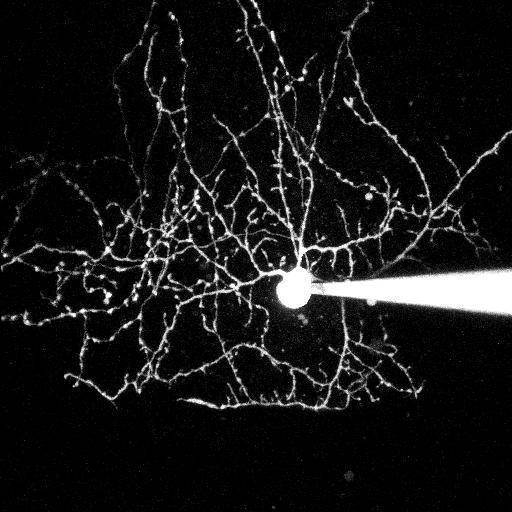
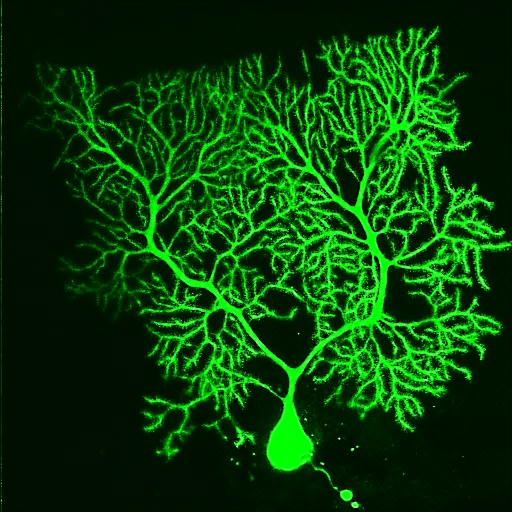
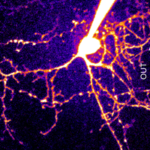
Synaptic basis of behavior A principal challenge in neuroscience research is to understand how the cells in the brain (neurons) use their specialized contacts (synapses) to route and transform information in order to perceive the world around us and in turn drive behaviors. One fascinating function of the nervous system is its ability to keep track of time. Sensations, thoughts, and actions are dynamic events that require the brain to encode the passage of time. For many tasks, such as playing music or sports, accurate execution requires the precise estimation of time intervals in the range of milliseconds to seconds. But how neuronal elements within brain circuits represent time is not understood. Synaptic connections between neurons change their strength dynamically during brief bouts of activity, and we hypothesize that they could therefore act like “cellular timers” and thus be a substrate for encoding time within neural networks to generate precise behaviors. A specialized region of the brain, the cerebellum, learns precise temporal details of our internal and external sensory world, to fine-tune motor and cognitive behaviors. Indeed, deficits of cerebellar function could account for altered sensory responses in schizophrenia or autism. Fortunately, the cerebellar circuit architecture is rather simple and has only a handful of well-defined neuron types. This makes it uniquely tractable to establish the role of each neuron type and its synaptic connections in generating precisely timed actions.
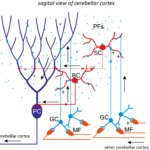
Hypothesis: The laboratory of Synapse and Circuit Dynamics (SCD) has made seminal discoveries about the various functions of synapses in the cerebellum (Figure 1) as well as the molecular organization within nerve terminals driving this diversity. Subsequently, we developed a mathematical model that makes predictions about how synaptic diversity is a substrate for circuit computations underlying animal behavior (Figure 2). The principal hypothesis is that dynamic changes in synaptic strength are necessary to generate a distributed representations of time, which can be used as a mathematical basis to learn arbitrary shapes of output neurons. This distributed representation of time enables the cerebellum to precisely time actions.
Approach: The SCD laboratory has implemented a multi-scale research program that links macromolecular organization at synapses to neural circuit function that drives well-timed behaviors. Projects in the laboratory include microscopy development, use of patch-clamp and dynamic two-photon imaging in acute brain slices, super-resolution imaging of synaptic macromolecular complexes, high-speed random access 2-photon imaging of neuronal population activity and single-unit recordings using high-density Neuropixels probes in awake behaving mice. Statistical and numerical methods are used to fit data sets to mathematically formalized hypotheses. 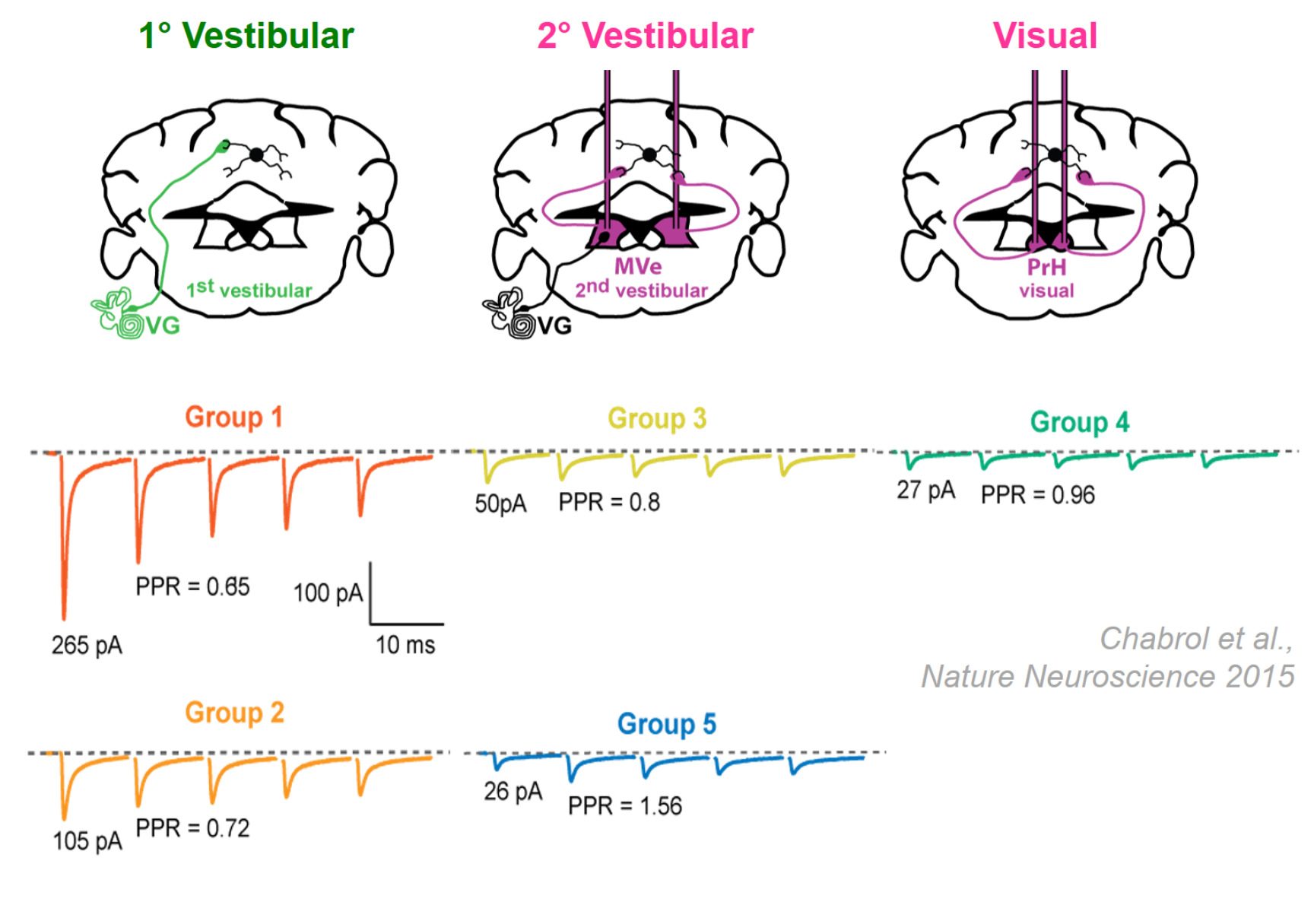
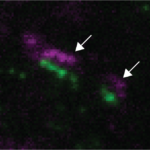
Figure 1. Diversity of the MF-GC synapse. Mean synaptic currents for five synapse types (groups) in response to 100 Hz train stimuli showing diverse amplitudes and short-term plasticity. 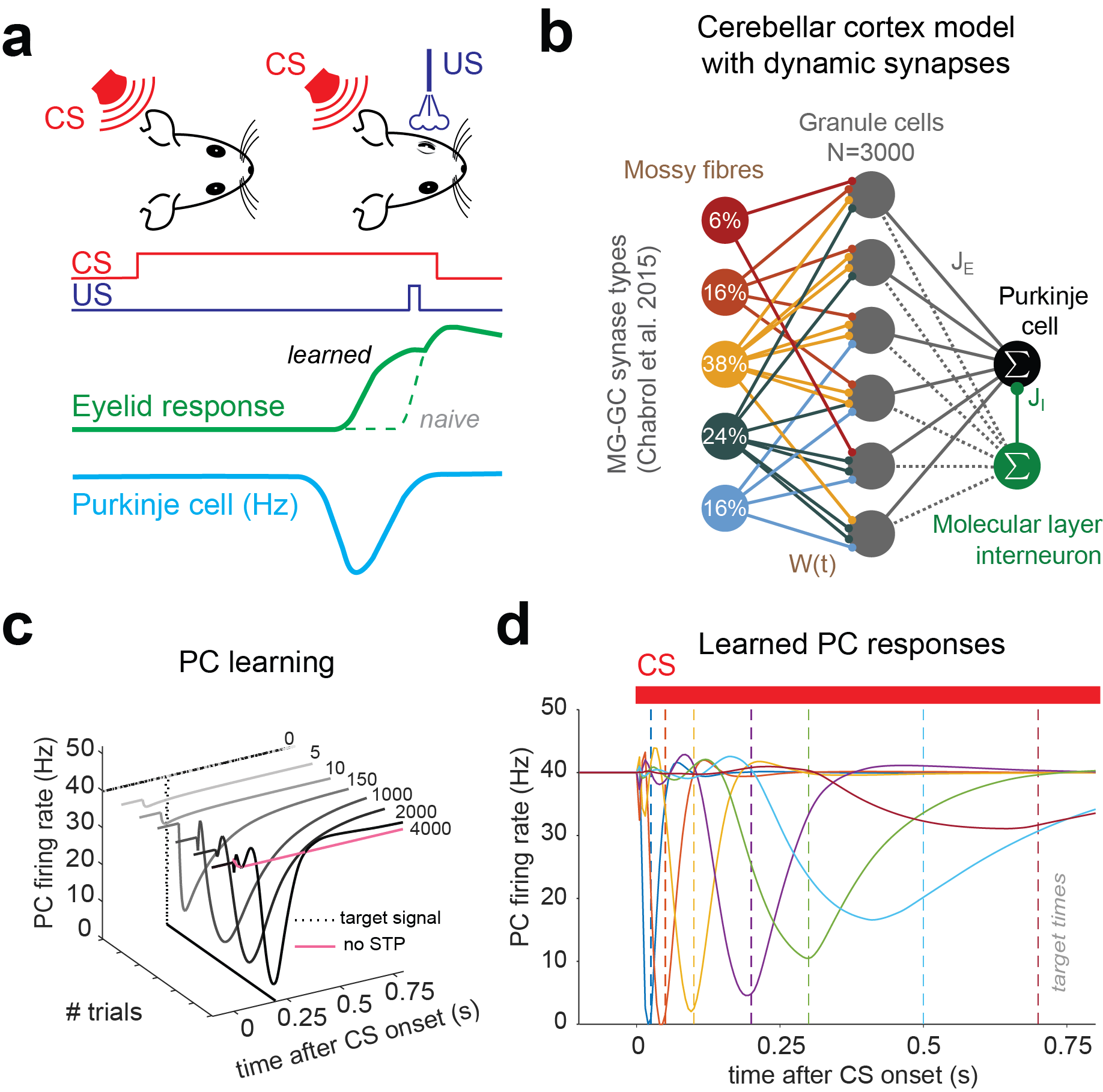
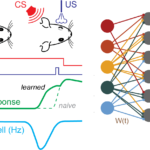
Figure 2. Simulating PC pauses during Eye-lid conditioning. a) Scheme of eye-lid conditioning. CS: conditioned stimulus (red). US: unconditioned stimulus (violet). After experiencing CS and US delivered at a fixed temporal contingency over many trials, the animal learns to close its eyelid before the US is delivered (green). A trough in PC activity(blue) precedes the eyelid closure (target time, grey dashed line). b) Scheme of cerebellar cortex rate model. MFs are classed according to synapse types from Chabrol et al. 2015. Percentages indicate relative frequency of MF groups. Insets: firing rate distributions for different MF groups. c) Example of eyelid learning over the course of 4000 learning steps for a 200 ms delay. Dashed line represents target signal used in supervised learning procedure. Without STP-induced GC transients, no PC trough can be learned (pink line). d) Eyelid learning for different target times. Different colors indicate PC responses after 4000 learning steps of distinct simulations and corresponding target times (dashed lines).
Positions available: Postdoctoral fellowship positions are available for the study of the synaptic basis of neural circuit computations underlying cerebellar-dependent behaviors. We have previously shown that the diversity of synaptic strength and plasticity is important for the temporal coding of multisensory stimuli. Using theoretical approaches (Figure 2) we now hypothesize that synaptic diversity is critical for the perception of temporal sequences of sensory stimuli and temporal learning. We will attack this problem using three strategies: 1) synaptic and neuronal mechanisms will be explored using imaging and electrophysiology approaches in acute brain slices, 2) computational modeling and statistical analysis, and 3) using high-density neural pixel recordings and state-of-the-art high-speed random access in vivo 2-photon imaging of newly developed activity reporters (Ca2+ and neurotransmitter), tools ideally suited for recording the high-frequency spike and synaptic activity of cerebellar neurons. The simplicity of the cerebellar cortical connectivity lends itself to the recording from each of five principle neurons in order to directly test network model predictions of the computations the circuit may perform. We are accepting applicants for each approach (in situ, in vivo, and computational). Thus applicants should have previous experience in electrophysiology, imaging, or in vivo recordings in awake behaving animals, and a good command of signal analysis. The laboratory is multidisciplinary with in situ and in vivo neurophysiologists, physicists (optics), and theoretical neuroscientists working in a highly collaborative environment. If interested, please send a CV and letter of motivation to david.digregorio@pasteur.fr.