Our research group focuses on the means to control chronic viral infections. We aim to define the properties of optimal antiviral T cells and to amplify these cells in immunotherapeutic approaches. Single T cell analyses offer the possibility to precisely define the nature of T cell receptors (TCRs) most apt at targeting persisting viruses. In addition, the reconstruction of human tissues in organ-on-chips now provides a mean to directly evaluate the capacity of antiviral T cells to reach and target infected cells in situ. We are building on these technologies to characterize T cells that can efficiently target two major viral pathogens, HIV-1 and SARS-CoV-2.
Research Axis 1: Characterization of shared T cell receptors in HIV controllers
Rare patients spontaneously control of HIV replication in the absence of antiretroviral therapy. These patients, called HIV controllers or Elite controllers, show signs of a particularly efficient antiviral immune response that contains HIV replication to very low levels. We have found that high-quality CD4+ T cell help is essential to maintain HIV control. In particular, CD4+ T cells from HIV controllers preferentially express shared TCRs of high-affinity that detect HIV Gag antigens with very high sensitivity (Benati, J Clin Invest 2016). The immune system is thus in constant alert, and can detect and respond to viral replication “blips” very rapidly to maintain viral control.
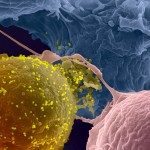
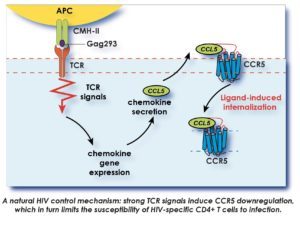
We have shown that the transfer of a high-affinity Gag-specific TCR to healthy donor T cells is sufficient to confer the efficient antiviral functions characteristic of controller CD4+ T cells, including the capacity to lyze infected target cells (Galperin, Sci Immunol 2018). Signaling through high-affinity Gag-specific TCRs also triggered an abundant secretion of beta-chemokines, which bound and internalized the HIV coreceptor CCR5, thus limiting the susceptibility of CD4+ T cells to HIV infection (Claireaux, Nat Comm 2022).
These findings open the possibility of TCR transfer applications, with the aim of establishing HIV control in patients who did not naturally control the virus. We propose to characterize the most prevalent TCRs involved in natural HIV control, an to evaluate their antiviral effects in TCR transfer models. We will also determine whether the TCR signatures associated to natural HIV control can be induced by candidate HIV vaccines.
Research Axis 2: Characterization of T cell responses in Long COVID
Long COVID is now recognized a major public health problem, as more than 10% of patients with SARS-CoV-2 infection show persisting symptoms more than 3 months after the acute infection stage. Long COVID patients suffer from a variety of persisting symptoms, in which debilitating fatigue, brain fog, and dyspnea predominate. The causes of this post-viral syndrome remain mostly unknown. In the PERSICOT project, we will test the hypothesis that an inefficient T cell response may allow the SARS-CoV-2 virus to persist at low level and cause inflammatory damage in Long COVID patients. The project is carried out in collaboration with the clinical team of D. Salmon-Ceron (AP-HP). The project involves the characterization of T cell responses to immunodominant SARS-CoV-2 epitopes, the characterization of the predominant TCRs in patients with resolved COVID and Long COVID, and a comparison of viral persistence and inflammation in both groups.
Research Axis 3: Development of organ-on-chips to evaluate antiviral strategies
Organ-on-chips represent 3D cultures of primary human cells that are continuously perfused with nutrient medium by a microfluidics system. Human cells are provided with a more physiological environment than in classical 2D cultures, and self-organize to reconstitute human tissues. Organs-on-chips provide new models to better mimic human physiology and help predict the effects of antiviral strategies.
We are developing a two-compartment lymphoid organ-on-chip (LO chip) based on the EmulateTM microfluidics technology. We found that the LO chip provides a highly sensitive system to measure the interactions between antiviral CD4+ T cells and memory B cells from volunteers who received a COVID vaccine. The LO chip will be used to compare different vaccine boosting strategies directed at containing SARS-CoV-2 variants and will be tested for its capacity to predict antigen immunogenicity.
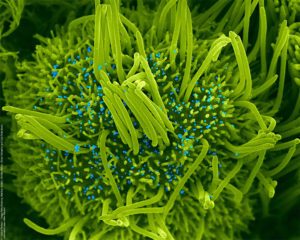
We are also using reconstructed airway epithelia to study the persistence of SARS-CoV-2 in its primary targets, the ciliated cells. We showed in this system that SARS-CoV-2 induces a loss of motile cilia in its target cells, which impairs the clearance of viral particles and may facilitate viral dissemination (Robinot, Nat Comm 2021). The image below shows a partially deciliated cells (green) with SARS-CoV-2 particles accumulating close to the plasma membrane (cyan).
We will further develop this system to generate an airway organ-on-chip that also includes a perfused vascular compartment. This system will be used to dissect the mechanisms of SARS-CoV-2 persistence and test the efficiency of antiviral molecules. The airway-on-chip will also be key to evaluate the capacity of TCR-transduced T cells cells to migrate into the epithelial layer and target SARS-CoV-2 infected cells. The project is done in close collaboration with the team of the Biomaterials and Microfluidics core facility of the Pasteur Institute.