Research in our lab strives to understand the mechanisms of the host immune response to human fungal pathogens, such as Candida albicans, Aspergillus fumigatus and Cryptococcus neoformans.
Combining human, murine, in vitro and in vivo experimentation models we are deciphering the immunological modulation of the innate immune response triggered by fungal infections and the associated innate immune memory. On the one hand we unravel the molecular mechanisms and physiological characteristics associated with the modulation of the innate immune memory. On the other hand we decipher the fungal associated molecules and factors that triggers such modifications of the innate immune system.
RESEARCH INTERESTS Fungal immune responses
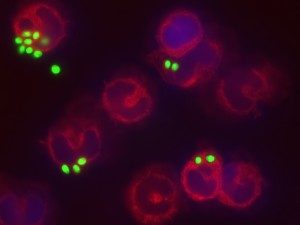
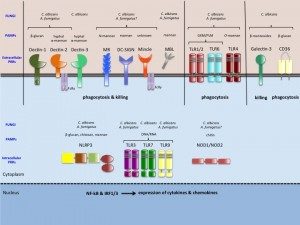
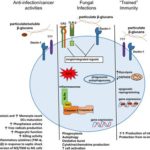
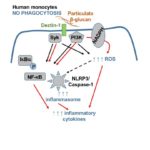
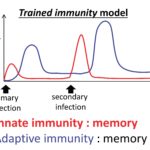
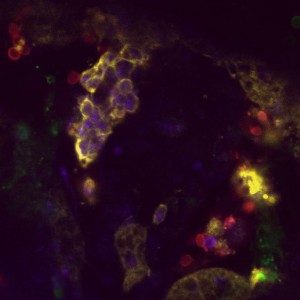
When PRRs bind fungal components, the pathogen-associated molecular patterns (PAMPs) which are often microbial cell surface components (mannans, beta-glucans etc…), they trigger intracellular signals resulting in phagocytosis, initiation of killing mechanisms (e.g. production of reactive oxygen species) and initiation of inflammation trough the production of cytokines, which ultimately would also help drive the development of adaptive immunity. Indeed, the innate immune response can be followed by an adaptive response, which is initiated by cells that present antigen of the fungus. Monocytes can rapidly differentiate into dendritic cells that transport ingested fungi to draining lymph nodes where they prime and expand fungal-specific T helper cells. Memory T cells and naïve T cells will be recruited and activated, and the cytokines profile present in the micro-environment will polarize the T helper lymphocytes response towards a predominant Th1, Th2 or Th17 response. Of note, monocytes can also increase the innate neutrophil conidiacidal activity and play a crucial role in controlling disseminated fungal infection. Innate immune memory The immune response is classically designated as innate or adaptive. Adaptive (or specific) immunity, requiring specialized cells such as such as T and B lymphocytes, specifically recognises a pathogen and builds immunological memory directed to that specific pathogen. Current vaccination strategies are based on the latter property of our immune system. In contrast to the adaptive immune response, current opinion holds that the innate immune response is non-specific and unable to build up immunological memory. However, the assumption that innate immune responses do not exhibit memory has to be challenged. Adaptive immunity-independent protection to re-infection is present in both plants and insects. These organisms lack specific adaptive immunity, but the state of their natural immunity can be enhanced by exposure to microbes and their products, which contradict the paradigm that innate immune responses cannot adapt. Similar observations are occurring in higher animals, i.e., organisms with an adaptive immune response. Groundbreaking studies in mouse demonstrated that NK cells possess adaptive immune characteristics. Our own recent results establish a degree of memory attributable to innate monocytes using in vivo murine and in vitro human models. We show that mice defective in functional T and B lymphocytes can be protected against re-infection with C. albicans in a monocyte-dependent manner. C. albicans and beta-glucans induce functional reprogramming of monocytes, leading to enhanced cytokine production in vivo and in vitro. Genome wide analysis show monocyte training by beta-glucans is associated with stable changes in histone methylation and acetylation. This beta-glucans-specific signature could be mirrored to the Escherichia coli/LPS-induced tolerance (one of the classic mechanisms to regulate over-exuberant inflammation tolerance is clinically associated with “immune paralysis” of monocytes/macrophages). In addition, beta-glucans-trained monocytes present a metabolic shift in glycolysis. And now? We are particularly interested in understanding the boundaries of immunologic memory, the role of innate immune plasticity during differential secondary infections. Specific projects will appear soon!