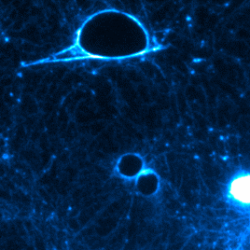
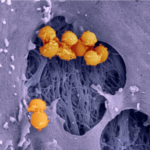
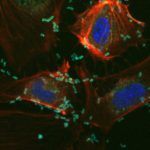
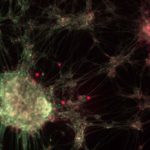
Bacterial protein toxins are remarkable protein nanodevices endowed with the capacity of inducing host specific unparalleled pathophysiological effects. Toxins offer a study subject of major interest for basic and medical research, as well as a critical topic for National security. Bacterial toxins under study in our laboratory include C. botulinum neurotoxins, that are among the most potent poisons in the world, while other toxins under study are associated with microbes responsible for highly frequent infectious diseases, such as C. difficile, S. aureus and uropathogenic E. coli (UPEC). UPEC and S. aureus represent a raising concern in term of acquisition of extended profiles of antibiotic resistance.
Our main research axes are linked to the characterization of the mechanism of action of protein toxins at the molecular level, defining how these toxins are delivered by bacteria and their route in cells/tissues, the prediction of virulence potential of existing and emerging pathogens by studying the distribution of these toxins in bacterial genomes, as well as the study of host cell intrinsic reactions to proteome and cellular architecture alterations triggered by these toxins.
Our work will address health issues related to the impact of asymptomatic carriage in the gut of toxin-producing bacteria on the development of age-related diseases and identify infection susceptibility genes. Moreover, we are conducting applied researches aiming at developing harmonized neurotoxin detection methods within the frame of a European consortium and develop antitoxin countermeasures.