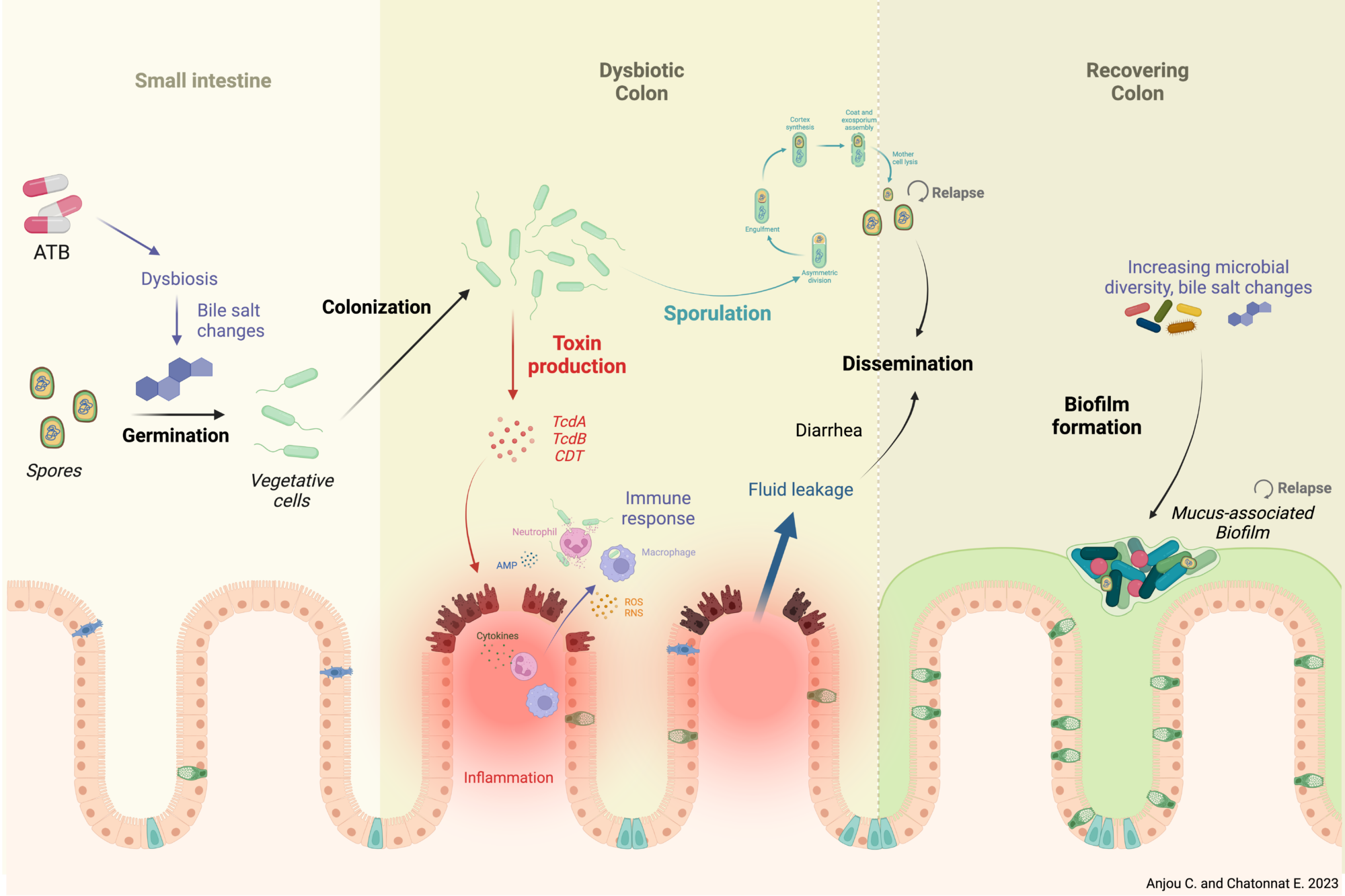
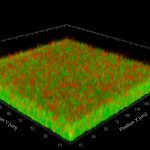
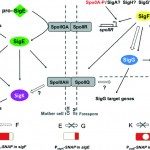
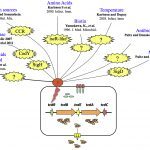
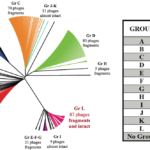
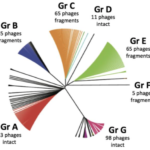
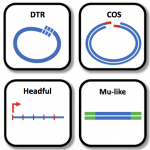
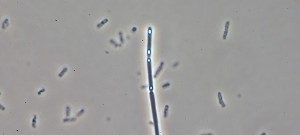
In Europe, the Gram-positive spore-forming anaerobic bacterium Clostridium difficile is the leading cause of intestinal nosocomial post-antibiotic infections in adults. The C. difficile infections (CDI) generally occur in patients with compromised gut microbiota. C. difficile is responsible for 15-25% of cases of antibiotic-associated-diarrhoea and for almost all pseudomembranous colitis cases. CDI is a multi-step process that includes i) oral infection by environmental spores of toxigenic C. difficile strains or caught off hospital staff, ii) spore germination in dysbiotic intestinal lumen, iii) gut colonization by C. difficile vegetative cells (VC) and iv) production of toxins responsible for the symptoms followed by the dissemination of C. difficile spores. The virulence of C. difficile is primarily due to the production of two cytolytic toxins: TcdA and TcdB, which exert deleterious effects on epithelial cells. However many virulence factors seem to be involved during the gut colonization and the process of CDI. Therefore, the aims of our research were to decipher the key steps of the C. difficile pathogenesis and particularly the intestinal colonization, which is an essential prerequisite before toxin production. This include i) the adaptive strategies used by C. difficile in response to stresses encountered during infection including mechanisms of resistance to oxidative/nitrosative stress and biofilm formation; ii) the complex regulatory network of C. difficile sporulation and the relationships that occur between spores and colonic cells during colonization, iii) the mechanisms of the regulation of toxin production in relation to cellular metabolic pathways and of their secretion and vi) the impact of prophages in the genetic variability of C. difficile strains.
Click to view graph
Connections
Members
Groups
Group
Group: Isabelle Martin-Verstraete

Isabelle Martin-Verstraete
Former Members
2000
2000
Name
Position
2015
2020
Emilie Camiade
Post-doc
2015
2020
Ana Antunes
PhD Student
2015
2020
Julio Aires
2015
2020
Laure Saujet
PhD Student
2015
2020
Frederic Barbut
Permanent Researcher
2015
2020
Thomas Dubois
Post-doc
2015
2020
Isabelle Poquet
Permanent Researcher
2015
2020
Pierre Boudry
PhD Student
2015
2020
Anna Maikova
PhD Student
2015
2020
Marc Monot
Head of Facility
2015
2020
Fanny Quenette
Undergraduate Student
2015
2020
Carolina Alves Feliciano
PhD Student
2015
2020
Elodie Cuenot
PhD Student
2015
2020
Audrey Hamiot
Research Engineer
2015
2020
Olga Soutourina
Permanent Researcher
2015
2020
Susana Matamouros
PhD Student
2015
2020
Nicolas Kint
Post-doc
2019
2023
Emile AURIA
PhD Student
2021
2023
Jazmin MEZA TORRES
Post-Doc