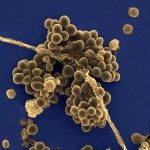
In the past five years, we have focused our research on Streptococcus gallolyticus subsp. galloltyticus (Sgg), an opportunistic pathogen causing septicemia and infective endocarditis in the elderly (above 65 years old). Intriguingly, this bacterium previously classified as Streptococcus bovis biotype I has been consistently and strongly linked to colorectal cancer (CRC). Our main goal is to determine whether the association of S. gallolyticus with colon cancer is causal or incidental, a long-lasting question in human medicine. This research project is currently supported by the National Institute of Cancer (INCA, PLBIO 16-025) and is performed in close collaboration with two partners, the Unit of Philippe Sansonetti (IP Paris) and the team of Catherine Robbe-Masselot, an expert in the field of glycobiology (University of Lille). Our current work is developping along the following lines: The first objective is to study the impact of Sgg on CRC development. Combining murine genetic models (Apc mutations) and in vitro cellular models (intestinal organoids ), we aim to answer the following questions: What is the impact of Sgg colonization on colonic mucins and its associated microbiota? Does the presence of Sgg induce oncogenic dysbiosis? Does the tumor constitute an entry portal for Sgg into the blood? What are the “signaling pathways” induced by Sgg in host cells that may sustain a direct role in the oncogenic process? By comparing normal physiological and tumor conditions, we hope to identify relevant parameters of the intestinal niche (e.g. metabolite profile, microbiota composition, mucus characteristics) that influence colonization capacities of Sgg. The second objective aims at studying the production, secretion and regulation of gallocin, a new bacteriocin allowing Sgg to kill closely related commensal bacteria in the murine colon under tumoral conditions (Aymeric et al., 2018). Purification of gallocin should enable us to identify “target” bacteria in the human microbiota using in vitro artificial colon models. Induction of gallocin transcription by specific tumor metabolites will be investigated in vitro and in vivo using luminescent bacteria and IVIS imaging. The third objective is to identify novel pathogenic traits of Sgg and to associate specific genomic traits with disease using comparative genomics. This collaborative work has begun using 40 Sgg isolates provided by Pr Claire Poyart, head of the CNR Streptococci (Hôpital Cochin) and NGS carried out at SCELSE Nanyang Technological University, Singapour. Finally, a long-term objective is to better understand the host immune responses in response to Sgg infections and how Sgg manipulates and evades them. In conclusion, increasing our molecular knowledge about the specific traits of S. gallolyticus that contribute to CRC initiation and progression should help in the development of new strategies for CRC diagnosis, treatment and prevention (for recent reviews, Pasquereau-Kotula et al., 2018; Aymeric et al., 2018).
Members
Former Members
2000
2000
Name
Position
2015
2019
Giulia Oliva
Post-doc
2015
2019
Mariana Martins
PhD Student
2015
2019
Noémi Réka Szili
PhD Student
2015
2019
Julian Lichtl-Häfele
Undergraduate Student
2015
2019
Maureen Micaletto
Undergraduate Student
2015
2019
Chloé Cassaro
Undergraduate Student
2015
2019
Ségolène De Champs
Undergraduate Student
2015
2019
Anne-Emmanuelle Roux
Undergraduate Student
2015
2019
Fanny Quenette
Undergraduate Student
2019
2020
Marta Garcia-Lopez
Undergraduate Student
2019
2020
Juliette Vadepied
Undergraduate Student
2017
2020
Alexis Proutière
PhD Student
2021
2021
Alexis Proutière
Post-doc
2021
2021
Paul Monassa
Undergraduate Student