Our work deals with some aspects of bacteria’s impressive capacities to acquire and exchange genes… which enable bacteria to evolve very fast and efficiently!
We study the mechanisms responsible of the bacterial genome variability, with a special interest for those involved in exogenous gene acquisition – the horizontal gene transfer. Our model system is the integron, a natural genetic engineering system involved in the development and dissemination of antibiotic resistance genes among Gram-negative species. We are characterizing the molecular mechanisms in these elements, as well as their evolution and phenotypic impact on host cell physiology.
We also study of the effects of subinhibitory concentrations of antibiotics, on the SOS response induction and beyond to their role as general stress inducers. We mostly use genetics and postgenomics approaches to tackle these questions.
In parallel we are investigating the factors playing a role in the repair and maintenance of the two chromosomes of Vibrio cholerae. All Vibrio species have genome constituted of two circular chromosomes with distinct dynamic characteristics, and we aim at understanding both the selective benefit of this organization as well as the specific machinery in charge of their maintenance.
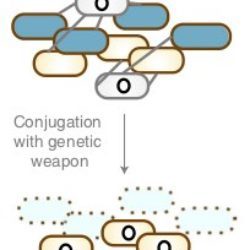
We recently developped a new antimicrobial technology based on the expression and delivery of specific toxin-intein modules which are only activated in the targeted pathogenic bacteria.
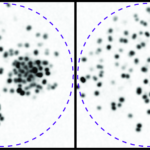
An illustration of our most recent article in Nature Biotechnology