About
A common phenomenon in most microbial infections is the attenuation of the host’s immediate immune response, allowing for long-term colonization. We and others have shown that many pathogens achieve this by manipulating the epigenetic regulation of the host. Epigenetics is the study of heritable phenotype changes that do not involve alterations in the DNA sequence but the control of the accessibility to the DNA sequence. Indeed, we demonstrated that pathogens either release factors that directly targets the host chromatin or hijack its epigenetic machinery, both resulting in alterations of the host epigenome. Our project is based on the hypothesis that inhibiting these phenomena will give an advantage to the host and thus facilitate the elimination of the microbe. Importantly, epigenetic modifications, such as DNA and histone modifications, are reversible and modulate gene expression without changing the DNA sequence. This makes them ideal drug targets. In cancer treatment, epigenetic regulators (enzymes and chromatin-binding proteins) are already validated drug targets, with several molecules approved for clinical use. In the context of infection, drugs targeting chromatin have been explored very little. TheraEPI proposes an innovative strategy to fight antimicrobial resistance (AMR) by chemically targeting epigenetic modifications. Our findings strongly suggest that this could be an innovative therapeutic strategy to counteract bacterial advantages and strengthen the host’s natural defences. By targeting the host, this strategy should also minimize the possibility of resistance emergence.
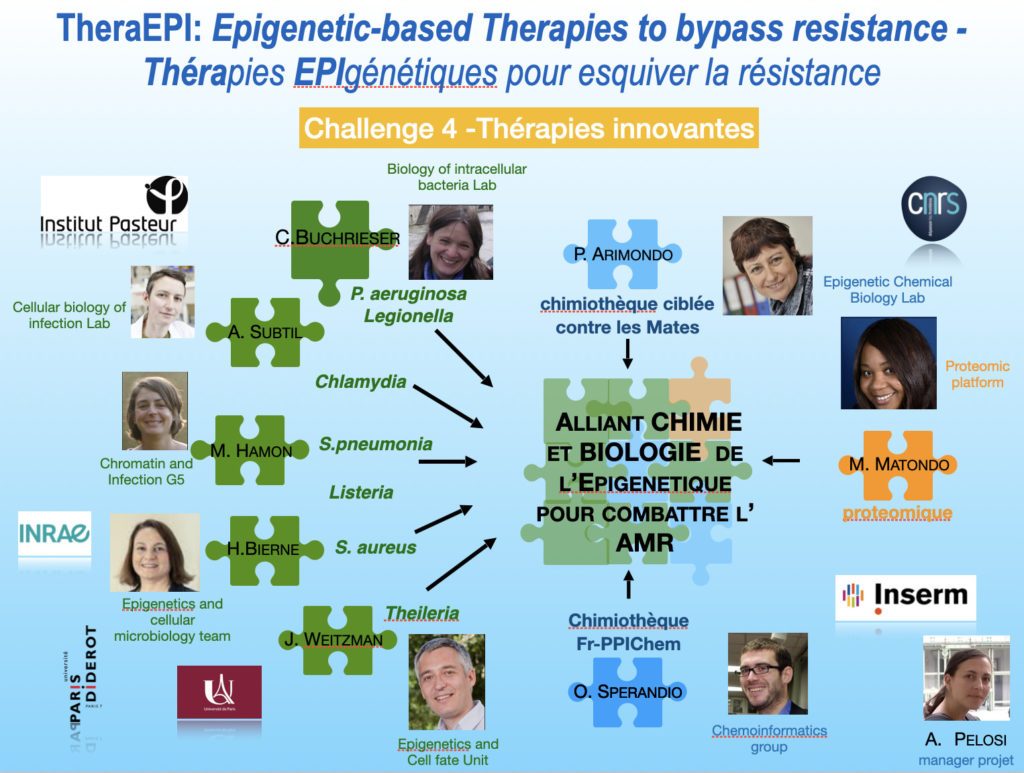
One of the strengths of the project lies in its interdisciplinarity since it combines expertise in the biology and epigenetics of infection (5 teams), in proteomics (1 team) and in drug design, chemo-informatics and medicinal chemistry of epigenetics (2 teams). We will explore seven pathogens (of the genera Chlamydia, Legionella, Listeria, Pseudomonas, Staphylococcus, Streptococcus and Theileria) as models of infection to identify new, specific epigenetic drug candidates and their targets.