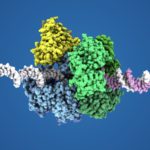
About
One fundamental challenge of cells is to accurately copy their genetic material for cell proliferation. DNA replication is performed by sophisticated multi-protein machines, which must synthesize DNA continuously and copy it accurately at the proper time in the life-cycle of the cell. Many of the replication factors that help replicate DNA act together in a dynamic complex with moving parts referred to as a replisome, loosely analogous to a sewing machine. However, our understanding of how these individual factors interact with one another during the replication processes is still fragmentary. Novel technical developments in Cryo-EM and single-particle analysis combined with protein crystallography and biophysical analyses, now provide biochemists with powerful tools to approach fundamental questions in DNA replication. We aim to approach fundamental questions in DNA replication by using hyperthermophilic Archaea as a model system. Determining the structure of the core replisome is required in order to understand how the leading-strand and lagging-strand DNA polymerases are coupled to the progression of the replicative helicase, as well as the structural basis for the repeated initiation on the lagging strand. We also aim to understand how the advancing replisome deals with obstacles, such as DNA damage, and how coordination is achieved between the moving replisome and DNA repair components during replication-coupled DNA repair. The expected results of this proposal will shed light on the origin of the replication system in the tree of life, including eukaryotes, and should be of great interest for a large community of molecular biologists; in addition it will help devise new biotechnological applications of these thermophilic enzymes.