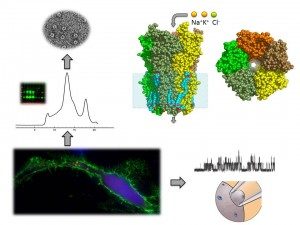
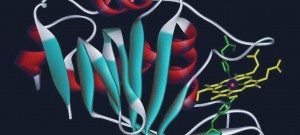
The cell membrane constitutes the main barrier for ion movement, and specialized proteins have been selected to catalyze the transport of various ions across the membrane: ion channels. Ion channels are integral membrane proteins (they cross the entire length of the membrane), and carry a hydrophilic water-filled pore that permit the passive flow of ions down their electrochemical gradient.
Our group is interested in a particular class of channels, which carry an internal gate undergoing opening/closing motions in response to ligands: Ligand-gated ion channel (LGICs). Among them, the pentameric LGICs (pLGICs) compose a large superfamily of phylogeneticaly-related membrane proteins, for which more than 40 genes are found in the human, accomplish a wide range of function in the cellular communication, particularly in neuronal communication. This superfamily includes nicotinic acetylcholine receptors (nAChRs), which mediate important pathways of cholinergic neuromodulation in the brain, GABA-A and glycine receptors, which mediate the majority of the inhibitory transmission in the brain and the spinal cord, as well as GABA-C and 5HT3 receptors. These proteins are involved in many human pathologies, and are the target of important therapeutic drugs including nicotinic derivatives, anxiolytics and anesthetics.
We aim at understanding, at an atomic resolution, the molecular mechanism governing the function of these proteins, and their regulation be therapeutic and addictive drugs.
We carry out a multidisciplinary approach: protein engineering, development of over-expression systems of these proteins in E. coli and cell lines to perform structural characterization by X-ray crystallography and cryo-electron microscopy, as well as functional studies by voltage-clamp and patch-clamp electrophysiology. To bridge the gap between structural and functional data, we also develop fluorescence techniques to follow the protein reorganization in real time and in physiologically relevant conditions. We also study and design drugs targeting these important receptors, combining electrophysiology and surface plasmon resonance.