About
Crossing of the epithelium and endothelium barriers by pathogens and their toxins represents essential steps in pejorative evolution of infections. We have discovered that several exotoxins from S. aureus and C. botulinum disrupt epithelium and endothelium barriers by targeting the small RhoA GTPase that governs actin cytoskeleton contractility. Our epidemiological analysis of S. aureus strains isolated from diabetic foot ulcers show a higher prevalence of edin in high-grade lesions. More generally, all of our work suggests that EDIN promotes S. aureus dissemination in tissues notably via the hematogenic route through a possible disruptive action of the endothelium barrier function. Consistently, ex vivo EDIN produces large holes in the endothelium of intoxicated arteries. These transendothelial macroaperture (TEM) tunnels also open in reaction to the rise of intracellular levels of cyclic-AMP broad cell signaling molecule that induces the edema toxin from Bacillus anthracis.
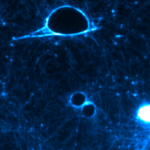
Opening of TEMs is a remarkable cell biology phenomenon that can be recapitulated in vitro by intoxication of endothelial cells. These large tunnels of 10-20 μm widen in 1-3 minutes, stabilize and close by extension of a membrane waves up to membrane resealing in about 10 minutes. By drawing an analogy with the physical process of dewetting of viscous liquids, coupled to atomic force microscopy, we have modeled in collaborations with Physicist at the Curie Institute and Pasteur Lille the formation of TEMs as a cellular form of liquid dewetting, i.e. nucleation and growth of dry patches in a viscous liquid film forced to spread on a non-wettable substrate.
The study of the dynamics of TEM in the frame of the model of dewetting represents a remarkable way to study quantitatively the coupling between mechanical forces, local recruitment of proteins that sense and deform membrane curvatures and the dynamics of the actin cytoskeleton. This shall highlight cellular mechanisms of mechanical adaptation of the endothelium to sheer stresses and infections. It should also provide important insights into general mechanisms of leukocyte transcytosis through the endothelial barrier.
Endothelial cell infected by EDIN-producing S.aureus
Endothelial cell intoxicated with the Edema toxin for Bacillus anthracis