Leptospirosis is a zoonotic bacterial disease found predominantly in impoverished populations inhabiting developing countries with tropical climates. Rodents are the main reservoir of the disease, excreting the bacteria in their urine. Humans are usually infected through contaminated water. Leptospirosis is estimated to cause more than one million severe cases with approximately 60,000 deaths per year. It is an emerging disease due to the growing number of inhabitants residing in urban slums and the increased frequency of extreme climatic events. Leptospirosis also affects animals worldwide, including livestock, and is therefore of economic importance. All control methods for leptospirosis implemented to date have been ineffective. The critical barrier to developing any effective interventions has been the limited understanding of pathogenesis of the disease. The “Biology of Spirochetes” Unit comprises the french National Reference Center for Leptospirosis and the WHO Collaborating Center for Research and Reference on Leptospirosis. The main missions of the NRC are to develop and improve diagnostic techniques, to perform epidemiological investigations, and to conduct the surveillance of leptospirosis in mainland France and in the French overseas territories (Martinique, Guadeloupe, Mayotte, etc). In mainland France, approximately 700 cases are diagnosed each year. In french overseas territories (New Caledonia, Martinique, Guadeloupe, Mayotte, La Réunion, French Polynesia, French Guyana), the incidence rate can be more than one hundred times that for mainland France. Since August 2023, confirmed and probable leptospirosis cases must be reported to the ARS (Agence Régionale de Santé) . Early laboratory diagnosis of leptospirosis is important to provide appropriate treatment of patients and to take rapid measures in case of an outbreak. Unfortunately, leptospirosis is often diagnosed late, due to its wide spectrum of symptoms, ranging from a flu-like syndrome to renal failure. The symptoms mimic the clinical presentations of
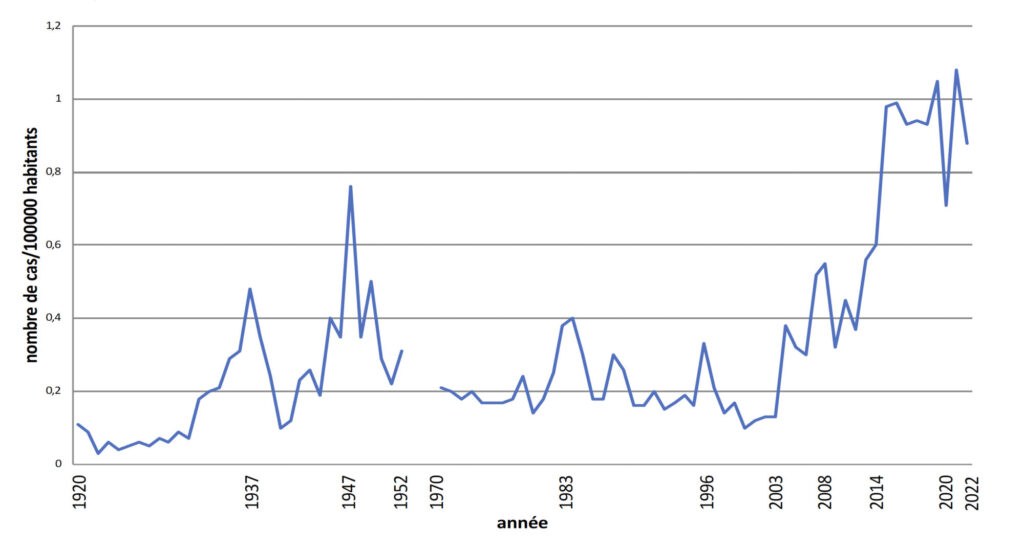
Fig. incidence of leptospirosis in mainland France.