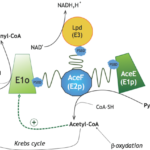
Nos activités de recherche sont orientées vers la caractérisation biochimique, biophysique et structurale de protéines impliquées dans la physiologie et la pathogénèse bactériennes, plus particulièrement dans les processus de signalisation.
Cliquez pour voir le graph
Connexions
Présentation
Membres

Pedro Alzari
Responsable de Structure
Responsable
Groupes
Group
Biologie structurale d’enzymes et complexes métaboliques

Marco Bellinzoni
Group
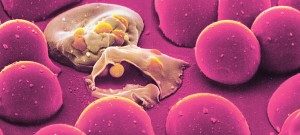
Biologie de Cibles du Paludisme et Antipaludiques

Jean-Christophe Barale
Anciens Membres
2000
2000
Name
Position
2015
2020
Bastien Sancerne
2015
2020
Natalia Lisa
Post-doc
2015
2020
Brigitte Vulliez-Le Normand
Research Engineer
2015
2020
Jocelyne Fraysse
Administrative Staff
2015
2020
Eduardo Bruch
Post-doc
2015
2020
Fredj Tekaia
Research Engineer
2015
2020
Lucile Le Chevalier-Sontag
Technician
2015
2020
Isabelle Miras
Technician
2015
2020
Gwénaelle Andre-Leroux
2015
2020
Tristan Wagner
PhD Student
2015
2020
Ariel Mechaly
Research Engineer
2015
2020
Jean-Michel Betton
Permanent Researcher
2015
2020
Pierre Vilela
Graduate Student
2015
2020
Jessica Rossello
PhD Student
2015
2020
Matteo Mori
PhD Student
2015
2020
Matteo Tassinari
PhD Student
2015
2020
Selma Topçu
Post-doc
2012
2020
Claudine Mayer
Professeur Université
2017
2022
Quentin Gaday
Post Doc / PhD Student
2020
2023
Emilie Yab
PhD Student
2021
2023
Daniela Megrian
Post Doc
2022
2023
Estelle Lecat
Technician
2022
2023
Johanna Gorkisch
Graduate Student
2022
2023
Antoine Gedeon
Post-doc
2023
2023
Antonina Gonet
Master Student