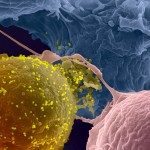
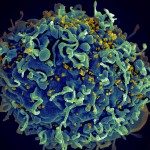
Our goal is to understand the mechanisms of effector immune responses to viruses and the related viral countermeasures, with a focus on antiviral antibodies. We combine basic and translational approaches to tackle novel questions on the mechanisms of action of antibodies against HIV-1, SARS-CoV-2 and emerging viruses.

Both SARS-CoV-2 and HIV-1 pandemics have highlighted the key contribution of antibodies to antiviral immunity. In the context of COVID-19, antibodies correlate to immune protection, are escaped by viral variants and are used as medicine. In the setting of HIV-1, the discovery of bNAbs have provided a framework for vaccine design and is raising new hope in the search for a cure. Still, many questions remain on the role of antibodies in antiviral immunity and how to optimize antibody-based therapies. How long antibody last after infection or vaccination and how this duration is regulated? What are the determinants of a broad antiviral response that resist viral diversification? What are the molecular mechanisms underlying viral control by antibodies? What are the viral countermeasures limiting antibody efficacy? How to optimize antibody-based therapies? I believe that a basic understanding of the mode of action of anti-SARS-CoV-2 and anti-HIV-1 antibodies will help answer these questions.
More precisely, our aims are:
- To decrypt the landscape of antiviral activities against SARS-CoV-2-infected cells and viral particles. Antibodies from COVID-19 patients who survived have a higher capacity to elicit ADCC than those from fatal cases, suggesting a protective action of ADCC. Likewise, the crystallizable fragment (Fc) region of antibodies is required for optimal therapeutic efficacy of monoclonal antibodies. However, the exact susceptibility of infected cells and the impact of non-neutralizing antibody activities, such as ADCC (antibody-dependent cellular cytotoxicity), CDC (Complement-dependent cytotoxicity) and ADP (antibody-dependent phagocytosis) on viral spread are ill-defined. Furthermore, how non-neutralizing antibody activities act against viral particles remains poorly characterized.
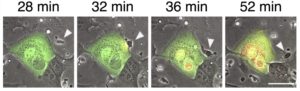
- To comprehensively evaluate COVID-19 therapeutic antibodies activities in patient samples to decrypt and survey their efficacy. Several monoclonal antibodies targeting the spike protein of SARS-CoV-2 are used in the clinic, but their efficacy is mitigated by variants. Evaluation of anti-S antibodies almost exclusively relies on in vitro neutralization data, limiting our understanding of their antiviral properties. Bio-disponiblity and inter-individual variability (for instance body mass index or pre-existing antibody levels) further influence clinical efficacy. In this translational work, we are collecting samples from patients receiving antibody therapies to investigate their efficacy against newly emerging variants, their mode of action in vivo and the reason underlying inter-individual variability in treatment efficacy.

- To investigate the capacity of anti-HIV-1 bNAbs to target the viral reservoir. Clinical and pre-clinical trials with HIV-1 bNAbs have revealed their capacity to target the viral reservoir. Thus, understanding the underlying mechanisms is critical to improve antibody-based strategies. Furthermore, it will open new avenues to understand the biology of the viral reservoir. Antibody may directly attack reservoir cells by ADCC. Therefore, we investigate the susceptibility of CD4 T cell subsets to ADCC, whether Env is expressed during homeostatic CD4 T cell proliferation and how ADCC limit viral reactivation. In addition to direct reservoir targeting, antibodies elicit a “vaccinal effect” that promote control. Thus, we investigate how dendritic cells and macrophages internalize immune complexes and prime CD8 T cells responses.
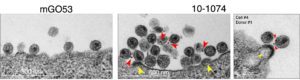
To achieve these aims, we use basic virology technics, culture systems of cell lines and primary human cells, live-imaging, high-content imaging, flow cytometry of cells and viruses and ultrasensitive protein quantification by digital ELISA. We work in collaboration with researchers and physicians inside and outside of the Institut Pasteur.
Overall, we are convinced that studying antibody functions will lead to exciting discoveries that will impact our understanding of immune responses and our use of antibodies in the clinic. Furthermore, given the key role of antibodies in antiviral immunity, a better understanding of their mode of action in the context of HIV-1 and SARS-CoV-2 is instrumental to rapidly fill the knowledge gaps that newly emerging viruses will create.