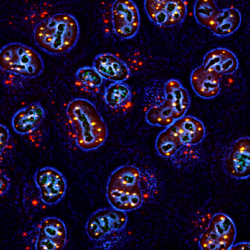
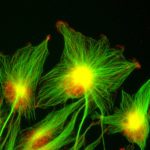
Our laboratory is interested in studying the epigenetic and cellular mechanisms underlying normal and pathological cell fate decisions with a particular emphasis on early embryo development and cancer.
How the identity of mature cells is maintained in multicellular organisms and how it is subverted in disease, particularly in cancer, are essential questions in biology and medicine. Cellular identity is dictated by transcriptional programs mediated by specific chromatin configurations, yet the mechanisms by which lineage choice decisions are choreographed to generate the adult organism or are corrupted in tumors remain poorly understood. Notably, the role of post-translational modifications of proteins by other proteins in this process is still enigmatic. We have shown recently that modification of chromatin proteins by the small SUMO protein acts as a general mechanism that safeguards cell identity. Lowering the SUMO epigenetic barriers facilitates cell fate change and is able to generate embryo-like structures from embryonic stem cells in a dish.
We are investigating the importance of SUMO modification in chromatin biology and gene expression and understanding how this modification regulates the establishment of the specific gene expression programs underlying cellular plasticity. We are using approaches including single cell -omics, chromatin biology, genome editing, protein biochemistry, live imaging as well as synthetic embryoid and organoid model systems. Beyond extending the mechanistic understanding of cellular diversification, we are probing the highly druggable SUMO pathway in the generation of faithful in vitro models of mammalian embryos and in malignancy reversion. In another approach, we are developing an integrated genome-wide profiling approach to dissect the molecular bases of hepatocellular carcinoma development in adolescents and young adults. Our research should bring new contributions in the mechanistic understanding of cellular diversity in normal and injured states, with a view toward therapeutic strategies seeking to manipulate sumoylation in regenerative medicine and cancer treatment.