Mission
Our core facility provides support in developing and characterizing VHH/nanobodies against the desired antigens. The main goal of this collaboration is to provide the right nanobodies that suit to the need of the user. We try not only to provide nanobodies but we can modify them: for example, according to the need, the nanobodies can be engineered to add a fluorochrome, to perform imaging; in this way, bispecific molecules can be created.
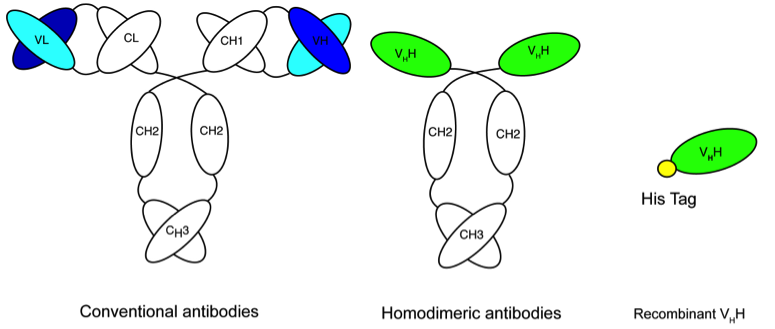
Nanobodies combine the advantages of binding activities of antibodies with properties of small molecules
Their features are
- small size : 15kDa (1/10th of the size of a conventional antibody)
- highly soluble
- high affinity for the antigen
- low immunogenicity (85-90% identity to human germline)
- highly stable
- well produced
- diffuse in tissues
- cross the Blood Brain Barrier
- Bind intracellular targets
Our expertise includes
- Phage display of phage VHH libraries
- Expression and characterization of nanobodies
- Site specific labeling with fluorochromes
- Automatization of the processes
Certifications and Networks
We are IBiSA labeled.
We are part of the EuroMabnet Network, CFL (Core For Life) and CTLS (Core technologies for life sciences).
To find out more about the Technology Department, see here.