About
Envelope glycoproteins of herpesviruses
Herpesviridae are a large family of double-stranded DNA viruses, counting more than 200 members that infect humans and a wide range of invertebrates and vertebrates. Herpesviruses carry a large linear DNA genome of 100 – 200 Kb, which is packed in an icosahedral capsid. The nucleocapsid is immersed in a protein rich matrix called tegument, which is enveloped by a lipid bilayer decorated with a dozen of surface glycoproteins involved in virus attachment and entry.
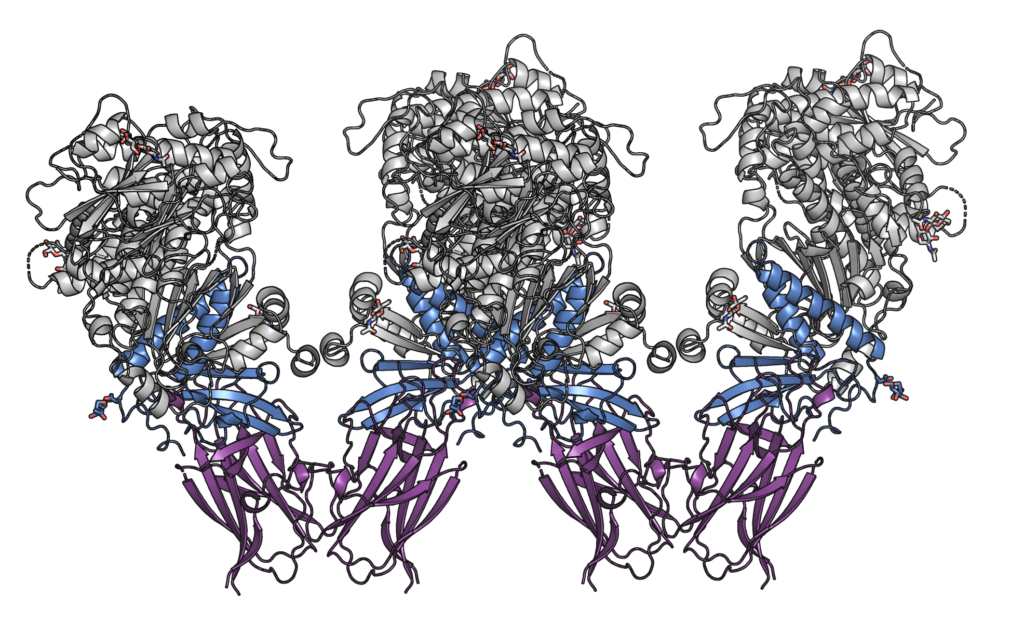
Herpesviruses enter cells by fusion of the viral and plasma membranes. Membrane fusion is triggered by binding of a viral envelope protein to a specific cellular receptor, and is catalyzed by a three-part fusion machinery composed of the envelope glycoproteins B (gB) and hetero-dimeric non-covalent complex made of glycoproteins H and L (gH/gL). The fusion apparatus of herpesviruses (gB-gH/gL) is conserved.
We are interested in understanding the molecular mechanism of herpesvirus entry -how does the binding to a receptor activate the fusion machinery, and how does a complex of gB and gH/gL mediate the merger of lipid bilayers. We are using a combination of biochemical, biophysical and structural biology methods to address these questions. One of the main goals is to determine three dimensional structures of the proteins involved in the entry process by X-ray crystallography. We are currently expressing a range of recombinant proteins from alpha-herpesviruses Herpes simplex virus 1, human herpesvirus 8, also known as Kaposi Sarcoma Associated Herpesvirus, and others.