About
Prion-like proteins spreading in neurodegenerative diseases (NDs): role of Tunneling Nanotubes (TNTs)
NDs are protein conformational disorders linked to the propagation of protein misfolding in the brain in a prion-like manner. A seminal discovery of the lab was that prion dissemination occurs by Tunneling Nanotubes (TNTs), a new mechanism of direct intercellular communication (Gousset, et al., Nature Cell Biol 2009).
In the last five years, we have further found that this mechanism is relevant for the intercellular spreading of other amyloid aggregates (Costanzo et al., JCS 201;, Abounit et al. 2016, EMBO J and Prion 2016, Loria et al., ACTA Neuropath., 2017) involved in neurodegenerative diseases including Huntington, Alzheimer and Parkinson.
We have proposed that TNTs are involved in the spreading of different amyloidogenic proteins in the brain and therefore represent a suitable target to halt incurable NDs (Victoria and Zurzolo, JCB, 2017).
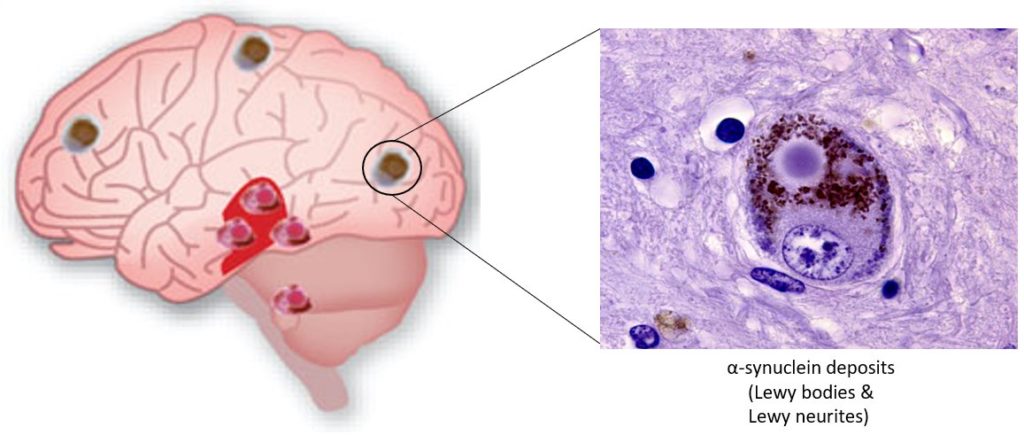
We also showed that stressed lysosomes are the vehicle of transfer of a-synuclein fibrils inside TNTs and allow, after fibrils escape, the seeding of cytosolic protein, thus explaining the progression of the pathology and highlighting an unsuspected role of lysosomes in this process. This is a new paradigm in NDs and opens new therapeutical perspectives (Victoria and Zurzolo, JCB, 2017).
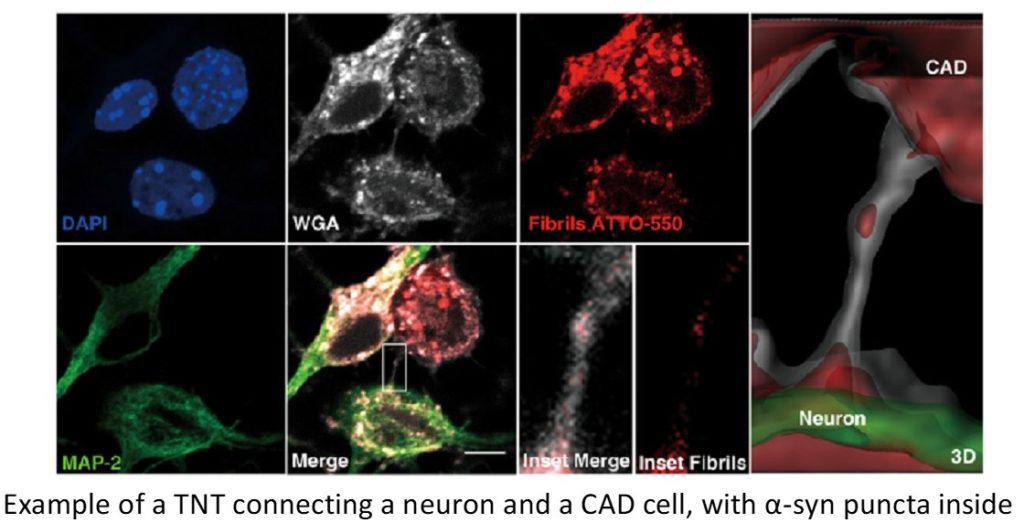
Lysosomal dysfunction is a common hallmark of NDs. We have shown that α-synuclein fibrils are transferred through TNTs between neurons inside lysosomes, and induce the misfolding/aggregation of the normal soluble protein (Abounit et al., EMBO J 2016). By applying high-content analysis and high resolution quantitative established in the lab we are now testing our groundbreaking hypothesis that dysfunctional lysosomes transferred through TNTs contribute to the propagation of the pathology. Specifically, we are studying: 1) the mechanism of lysosomal dysfunction mediated by α-syn fibrils in different neuronal cultures (cell lines, primary neurons and brain slices), 2) the mechanism of α-syn escape from lysosomes and 3) the mechanism of lysosome transfer in TNTs.
This project will decipher the contribution of TNTs and lysosome dysfunction in the progression of α-syn and tau pathologies. It will provide relevant translational information for innovative therapies in Parkinson and other neurodegenerative diseases.
“This project has received funding from the European Union’s Horizon 2020 research and innovation programme under the Marie Skłodowska-Curie grant agreement No 897378”