I am working on the Molecular Biophysics platform (PFBMI)(see https://research.pasteur.fr/en/team/biophysics/).
I got two main tasks in my work:
I collaborate with research teams of Institut Pasteur and other research centers on the characterization of the mass, size, shape, and stoichiometry of their biomolecules. To solve these questions, I use several and complementary approaches such as analytical ultracentrifugation (Optima AUC and Proteome Lab XLI), dynamic light scattering (DynaPro PlateReader), SEC-MALS, and Taylor Dispersion (Viscosizer TD). This task is tandem with my colleague Bertrand Raynal which can provide other techniques such as SAXS. As some questions are more challenging than others, I implement new experimental strategy using my expertise. This added value is recognized in the scientific paper that I have co-authored (see publication section). contact: biophysique@pasteur.fr
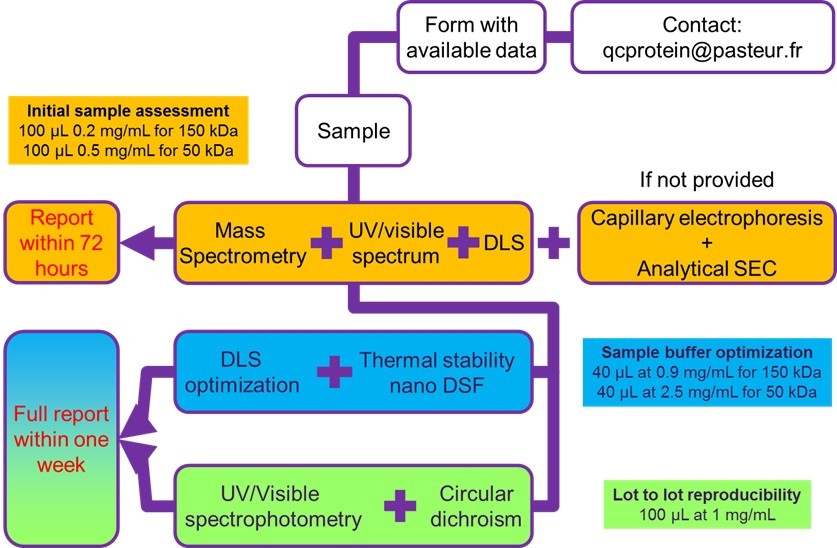
The second task is to run the quality control of purified proteins service. This service was officially launched in 2017 to help the research teams of the campus that had troubles in purifying their protein of interest and especially had difficulty to complete their downstream applications (Cryo-EM, X-Ray crystallography, activity tests, etc.) I have set up an experimental workflow that allows research teams to obtain a full qualitative control (QC) of their purified proteins and to help them to identify conditions in which their proteins is best behaved. In 2018, around 250 proteins were analyzed. Importantly, this service has become a transversal service with the protein production platform and offer optimization approches for nanoimaging (https://research.pasteur.fr/en/team/nanoimaging/)
For more information, please send an email to qcprotein@pasteur.fr
As a third task, I work on development of new methodology aquired by the molecular biophysics core facility. Recently, I have worked on the determination of intrinsic viscosities of biomolecules using the Viscosizer TD (publication in progress).