Présentation
Hepatitis C virus (HCV) infection can trigger the development of autoimmune disorders among which, cryobulininemic vasculatitis is the most prevalent. HCV-associated cryobulininemic vasculatitis is mediated by mono-/oligo-clonal IgM cryoglobulins with rheumatoid factor activity, thought to be produced by atypical CD21–CD27+ memory (AtM) B cells. AtM B cells are indeed frequently detected in patients with HCV-associated cryoglobulinemia vasculitis (HCV-CV) but are anergic, and prone to apoptosis.
Thus, we investigated the cellular and molecular processes by which abnormally expanded AtM could participate to HCV-associated autoimmunity. In collaboration with Pr David Saadoun (La Pitié-Salpétrière Hospital, INSERM UMR_S 959), we studied the effects of TLR9 stimulation in HCV-CV AtM B cells and found that it induced: (i) the secretion of TNFα and IgMs with rheumatoid factor activity by AtM; (ii) an AtM-specific transcriptional signature centered on TNFα overexpression; (iii) the stimulation and secretion of IFNγ and TNFα by effector T cells and conversely, a reduction of regulatory T-cell proliferation. Moreover, we showed that AtM B-cell expansions displayed intraclonal diversity of mutated IgM with features of antigen-driven maturation. AtM antibodies did not react against ubiquitous autoantigens or HCV antigens but rather, possessed rheumatoid factor (RF) activity with unique targeted epitopes on the human IgG Fc region.
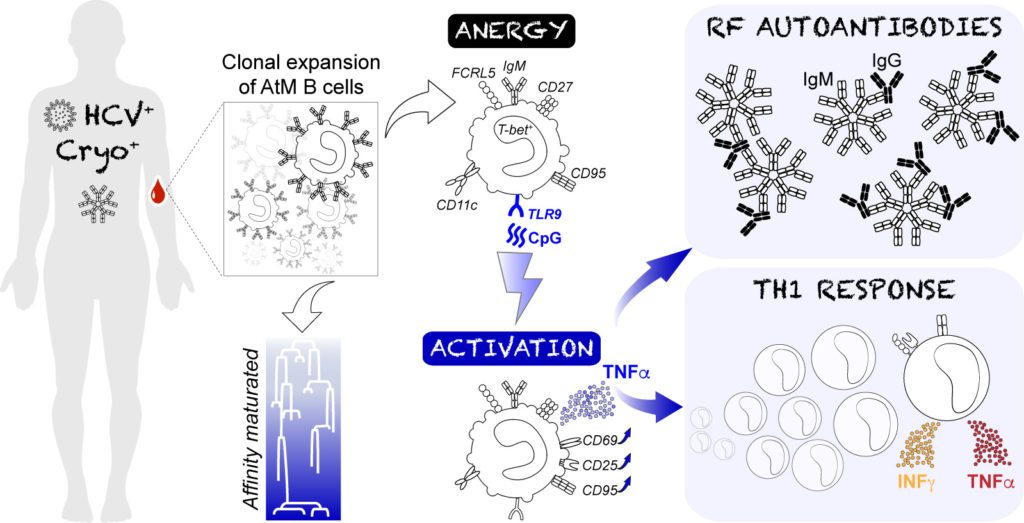
Thus, our data suggest a central role of memory B cells in the defective tolerance characterizing HCV-CV patients, via the TLR9-induced reactivation of anergic AtM, which eventually mediate RF IgM autoantibody and pro-inflammatory Th1 responses (Comarmond*, Lorin*, Marques*, et al 2019).