About
Correlative Light & Electron Microscopy (CLEM) approaches are methods of choice to image rare or dynamic events at high-resolution by Electron Microscopy (EM). The functional and dynamic information coming from Light Microscopy (LM) can be directly linked to the higher resolution information provided by EM. The combination of LM and EM imaging methods used depends on the question asked and on the sample involved.
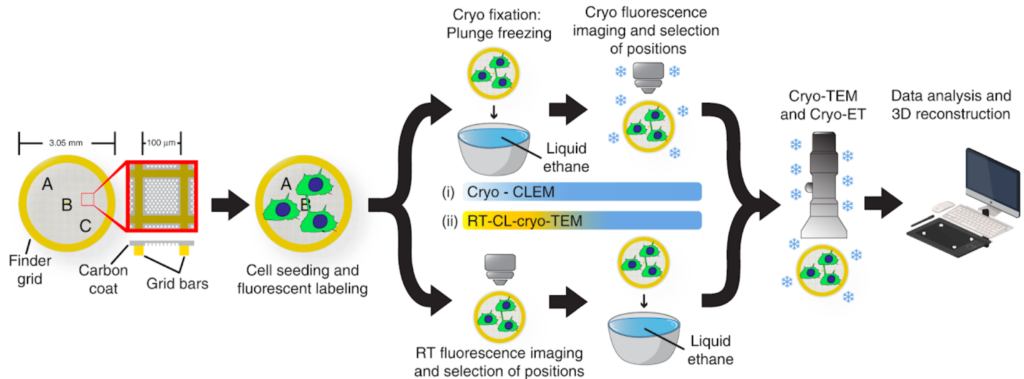
For our users we offer two main approaches: ‘Classical CLEM‘ where the LM imaging takes place before processing the sample for EM imaging and ‘High Precision on-grid CLEM’, where the sample is imaged both with LM and with TEM directly on the EM grid.
High Precision on-grid CLEM
For ‘on-grid CLEM’ approaches, samples that are typically prepared for TEM (particles, sections, entire cells) for room temperature or cryo, can be imaged prior TEM in a typical Light Microscope or in UBI’s cryo-light microscope (Leica EM Cryo-CLEM microscope). Full grid fluorescence maps can be acquired and positions of interest (POIs) recorded by LM. Fluorescent maps and POIs can then be registered to the TEM’s stage coordinate system and POIs can then be imaged at higher resolution. Specific fiducials (e.g. gold or fluorescent nanoparticles) can be used for high-precision CLEM at higher TEM magnifications.
Sartori-Rupp A, Cordero Cervantes D, Pepe A, Gousset K, Delage E, Corroyer-Dulmont S, Schmitt C, Krijnse-Locker J. & C. Zurzolo, (2019). Correlative cryo-electron microscopy reveals the structure of TNTs in neuronal cells. Nat. Comm. 10, 342.
Classical CLEM
In classical CLEM approaches samples are deposited or grown on special supports with integrated landmarks (e.g. MatTek dishes) and imaged by LM, live or chemically fixed, following fixation protocols that preserve at the same time sample’s ultrastructure and fluorescent signal. The sample is then further processed for EM, followed by EM acquisition and analysis. This method can be used for CL-TEM, for CL-SEM – in combination with gold immunolabelling – and for CL-FIB/SEM (Volume CLEM).
Mostowy S, Bonazzi M, Hamon MA, Tham TN, Mallet A, Lelek M, Gouin E, Demangel C, Brosch R, Zimmer C, Sartori A, Kinoshita M, Lecuit M, Cossart P. 2010. Entrapment of intracytosolic bacteria by septin cage-like structures. Cell Host Microbe 8:433-44.

Campbell-Valois FX, Schnupf P, Nigro G, Sachse M, Sansonetti PJ, Parsot C (2014). A fluorescent reporter reveals on/off regulation of the Shigella type III secretion apparatus during entry and cell-to-cell spread. Cell Host Microbe 15(2):177-89.
Access Protocol
….Under Construction….