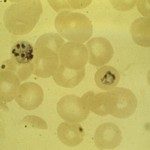
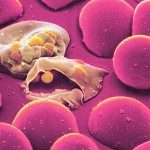
Malaria is a disease that continues to cause tremendous suffering and mortality in the tropics. This disease is caused by Plasmodium parasites. The species Plasmodium falciparum causes the severest and most deadly form of malaria. Scientists from Institut Pasteur studied the blood stage of the parasites and found a mechanism which could lead to new and efficient drugs against Malaria.
Despite sustained control efforts over the last 20 years, the WHO reported in 2020 that malaria cases remain high, with 241 million cases reported each year and an estimated 627 000 deaths. The main concern in the fight against malaria is the increasing resistance of parasites against the frontline drug, named artemisinin. Identification of novel drugs that overcome drug resistance is pivotal to enable progress towards malaria elimination.
The parasite enters the bloodstream during a mosquito bite and then multiplies in the liver: this is called the liver stage. After multiplying, the parasite goes back into the blood, invades and multiplies within host red blood cells: this is called the blood stage. “All the clinical symptoms of malaria are attributed to the blood stage. As the parasite multiplies within the red blood cells, it internalises and digests hemoglobin to provide amino acids for synthesis of parasite proteins. Digestion of hemoglobin leads to severe oxidative damage to membranes of the parasite” explains Chetan Chitnis, Head of the Malaria Parasite Biology and Vaccines Unit at Institut Pasteur. The parasite is surrounded by a membrane that protects it from the outside, therefore the repair of oxidative damage to membranes is essential for its survival.
Matthias Wagner, a PhD student working with Chetan Chitnis, has found that a human antioxidant protein called PRDX6, found in red blood cells, is taken up by the parasite along with hemoglobin. “PRDX6 is known to play a role in repair of oxidised membrane lipids. We found that the inhibition of human PRDX6 with an inhibitor called Darapladib is lethal for growth of P. falciparum blood stage parasites. Interestingly, co-treatment of P. falciparum with Darapladib and artemisinin also reduces the survival of artemisinin-resistant parasites that are not killed by artemisinin alone” says Matthias Wagner.
Targeting a human protein instead of a parasite protein for development of anti-malarial drugs is attractive because parasites cannot mutate the target gene to attain resistance. Development of PRDX6 inhibitors as novel drugs against malaria could play a critical role in eradication efforts in the long term.
This research has benefited from funding from Agence Nationale de la Recherche (ANR) and PasteurInnov. Matthias Wagner was part of the Pasteur-Paris University International PHD Program and Chetan Chitnis is a member of the ParaFrap Laboratory of Excellence. The researchers collaborated with several Institut Pasteur platforms including the electron microscopy Ultrastructural BioImaging platform, the Central Animal Facility and the Image Analysis Hub as well as colleagues in a number of other research institutes in Germany, Singapore and USA.
https://www.pasteur.fr/en/research-journal/news/new-drug-target-against-malaria