Click to view graph
Connections
Click to view timeline
Timeline
Projects
Tools
Publications
Download-
2023Analysis of in-patient evolution of Escherichia coli reveals potential links to relapse of bone and joint infections., J Infect Dis 2023 Dec 2:jiad528.
-
2017Long-term stability of gentamicin sulfate-ethylenediaminetetraacetic acid disodium salt (EDTA-Na2) solution for catheter locks, J Pharm Anal. 2018 Dec;8(6):386-393. doi: 10.1016/j.jpha.2017.09.004. Epub 2017 Sep 25..
-
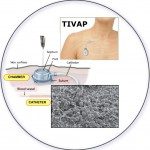
2017Central venous catheters and biofilms: where do we stand in 2017?, APMIS. 2017 Apr;125(4):365-375. doi: 10.1111/apm.12665..
-
2017Understanding biofilm formation in intravascular device-related infections, Intensive Care Medicine. Mar;43(3):443-446. doi: 10.1007. [Epub 2016 Aug 6].