Présentation
The identification of variants in DHX37 as a cause of 46,XY DSD opens up the possibility that some forms of human sex-reversal are ribosomopathies. Pathogenic variants associated with a lack of testis-determination or, the inability to maintain testis identity are clustered within the functional RecA1 and RecA2 domains of the helicase and involve amino acids that are conserved through to yeast (Figure 2a). Consistent with a role in early testis development, DHX37 is expressed specifically in somatic cells of the developing human, mouse and goat testis (Figure 2b).
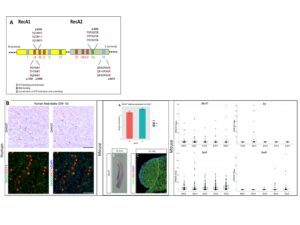
The identification of DHX37 as a component of the mammalian SD pathway forces a paradigm shift from the prevalent transcription-factor centered view. The mechanism(s) of how a mutation in a widely expressed protein involved in a basic cellular function can generate a highly-specific human phenotype is unknown. We are currently assessing the impact of DHX37 mutations on human ribosome biogenesis and identifying functionally relevant partners (RNA and protein) for DHX37 that are involved in SD.