Présentation
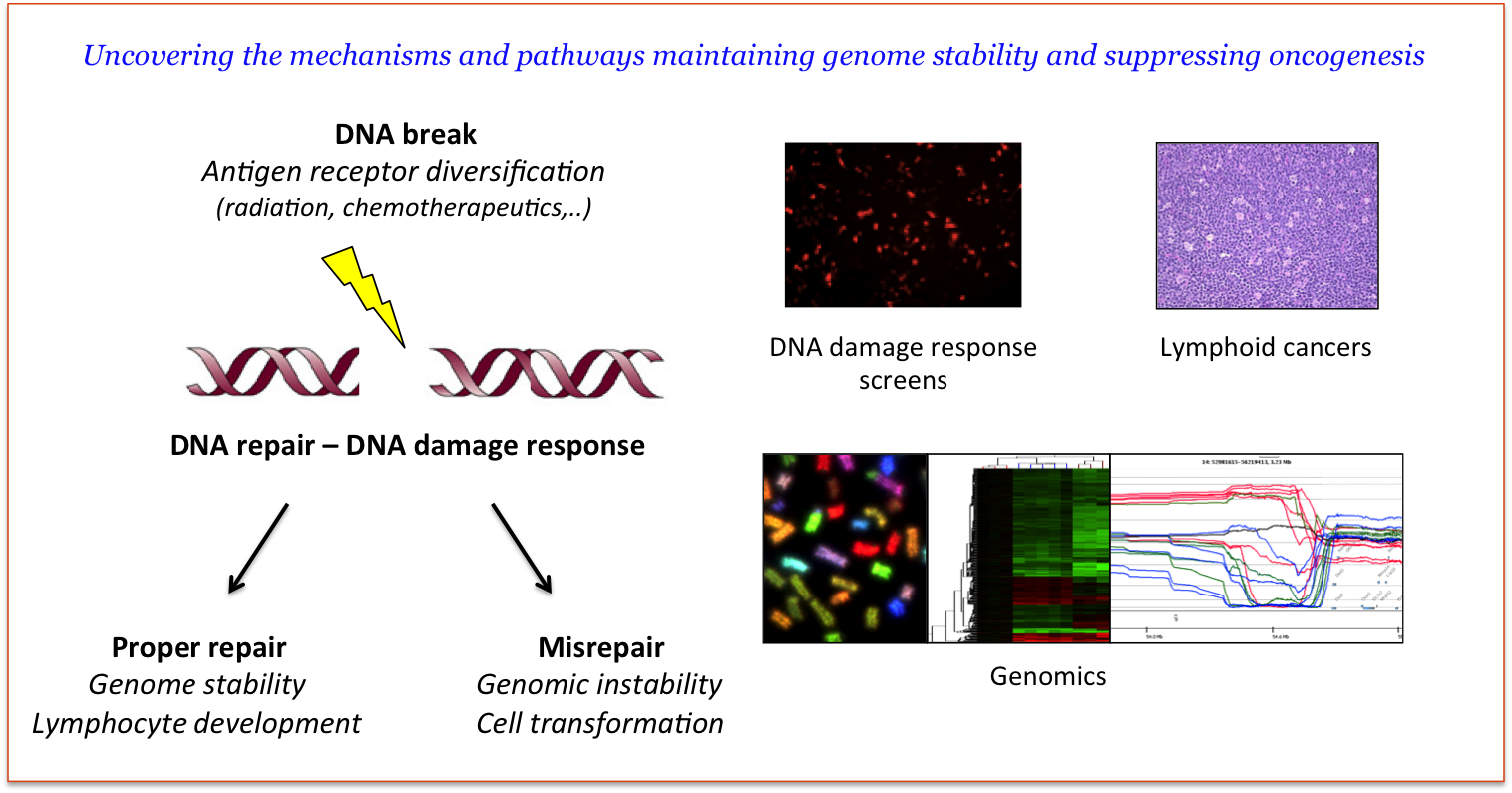
B and T cell lymphoid cancers are among the most common human malignancies. Many factors, endogenous and exogenous, are implicated in the etiology of these disorders, but key oncogenic lesions often arise through chromosomal translocations involving antigen receptor loci. These events frequently shows signs of having originated through a variety of errors in RAG1/2 protein-mediated V(D)J recombination. While there are several theoretical possibilities to explain various mechanisms through which such errors occur – lymphocyte development arrest at stages of antigen receptor diversification, recombination errors due to aberrant RAG-, DNA repair-, DDR-activities, etc. –, there has yet to be functional cancer model organisms to test these hypotheses. We use a series of mouse models harboring defects in DNA double strand break regulation, and as a consequence readily develop B- and T-cell tumors. These lymphomas contain numerous genomic aberrations reminiscent of human malignancies. We apply a combination of genomic, transcriptomic, computational and genetic screening approaches to understand the basic nature of genome instability in developing lymphocytes and to identify the genomic lesions and oncogenic pathways underlying lymphomagenesis. From a general perspective, our studies will provide new insights into the mechanisms of DNA rearrangements, DNA repair, genome stability and tumor biology.