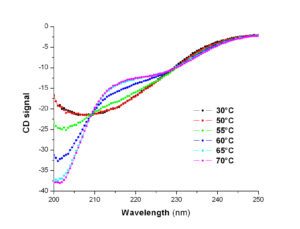
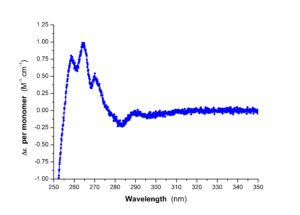
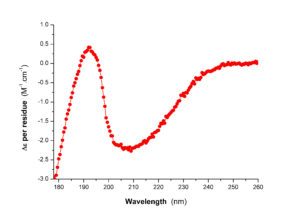
The PFBMI has an J-1500/ 450W instrument. Studying macromolecules (and in particular proteins) by circular dichroism allows to obtain information about their folding. For proteins, far UV (180-260 nm) and near UV (250-330 nm) circular dichroism measurements give insight respectively into their secondary structure content and their tertiary organization. The thermal stability of folding can be determined by applying a temperature gradient to the sample. Similarly, it is possible to quantify the conformational stability of a molecule at constant temperature or the structural changes induced by the formation of a complex, by performing a titration, respectively by a denaturing agent or a ligand.
| Circular dichroism in the near UV | Circular dichroism in the far UV |
 |
 |
Thermal stability of folded molecules can be determined by performing temperature scans. Similarly, conformational stability of a molecule and structural changes induced by complex formation can be checked at constant temperature by adding step by step chemical denaturating agents or specific ligands using a computer driven titrator.