Link to Pubmed [PMID] – 19395494
J. Bacteriol. 2009 Jul;191(13):4195-206
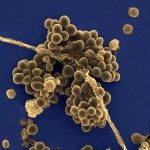
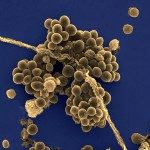
In streptococci, the secA2 locus includes genes encoding the following: (i) the accessory Sec components (SecA2, SecY2, and at least three accessory secretion proteins), (ii) two essential glycosyltranferases (GTs) (GtfA and GtfB), (iii) a variable number of dispensable additional GTs, and (iv) a secreted serine-rich LPXTG protein which is glycosylated in the cytoplasm and transported to the cell surface by this accessory Sec system. The secA2 locus of Streptococcus agalactiae strain NEM316 is structurally related to those found in other streptococci and encodes the serine-rich surface protein Srr1. We demonstrated that expression of Srr1 but not that of the SecA2 components and the associated GTs is regulated by the standalone transcriptional regulator Rga. Srr1 is synthesized as a glycosylated precursor, secreted by the SecA2 system, and anchored to the cell wall by the housekeeping sortase A. Srr1 was localized preferentially at the old poles. GtfA and/or GtfB, but not the six additional GTs, is essential for the production of Srr1. These GTs are involved in the attachment of GlcNac and sialic acid to Srr1. Full glycosylation of Srr1 is associated with the cell surface display of a protein that is more resistant to proteolytic attack. Srr1 contributes to bacterial adherence to human epithelial cell lines and virulence in a neonatal rat model. The extent of Srr1 glycosylation by GtfC to -H modulates bacterial adherence and virulence.