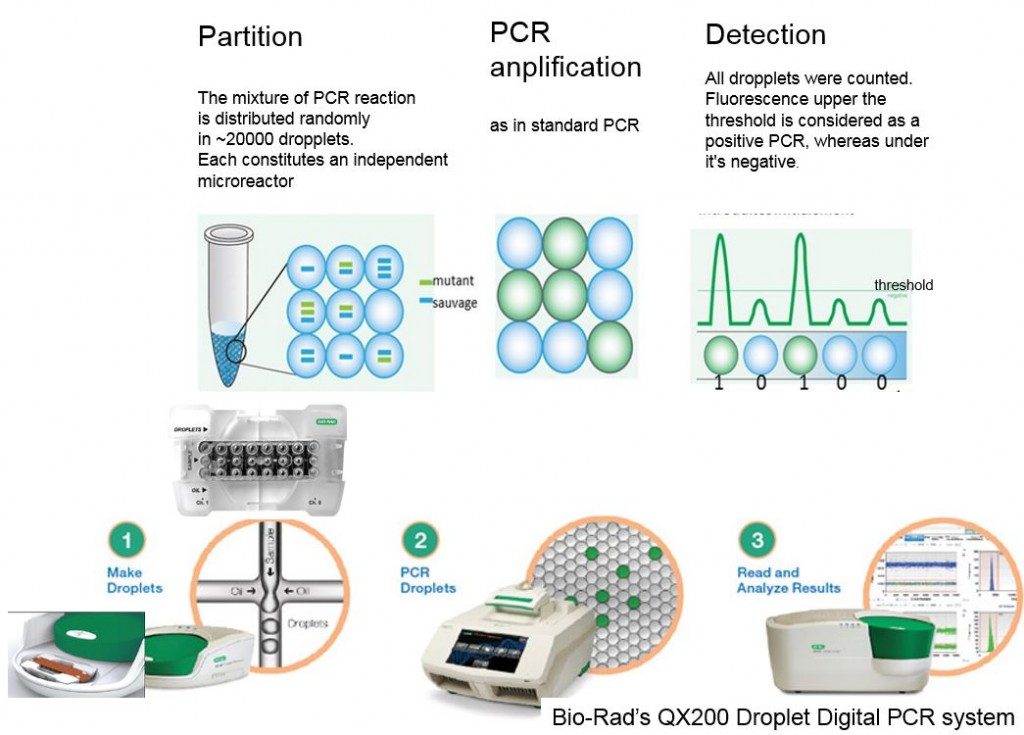
Présentation
The digital PCR is a new manner to quantify DNA molecules by PCR.
Because the digital PCR interrogates the number of molecules at the end of the 40 cycles of the PCR (end-point analysis) this quantitative method is thought to be more robust than the real-time-PCR by being less sensitive to the PCR efficiency (possibility to use longer amplicons and less sensitive to poisons).
The digital PCR allows to quantify very low number of DNA copy (by decreasing the complexity of the samples) and it allows theoretically to measure differences as small as 10% with high reliability.
The digital PCR is also interesting because it provides an ease to design PCR of multiple targets in a single assay (multiplexage with or without Taqman probes). The QX200 apparatus works with PCR mix with Taqman probes, or with EvaGreen. This last mix works with classical primers designed for real-time PCR.
However, due to the relative weakness of the measurement range (3 LOG), this approach requires to pre-estimate the DNA samples using for instance the conventional real time PCR.
The digital PCR works by diluting a sample into many partitions (droplets) and counting the number of partition in which a reaction occurs. The strategy is divided to conquer.