Lien vers Pubmed [PMID] – 27586688
Lien vers HAL – pasteur-01377851
Lien DOI – 10.1038/ncomms12629
Nature Communications, 2016, 7, pp.12629. ⟨10.1038/ncomms12629⟩
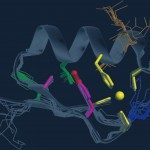
The NF-kB pathway has critical roles in cancer, immunity and inflammatory responses. Understanding the mechanism(s) by which mutations in genes involved in the pathway cause disease has provided valuable insight into its regulation, yet many aspects remain unexplained. Several lines of evidence have led to the hypothesis that the regulatory/sensor protein NEMO acts as a biological binary switch. This hypothesis depends on the formation of a higher-order structure, which has yet to be identified using traditional molecular techniques. Here we use super-resolution microscopy to reveal the existence of higher-order NEMO lattice structures dependent on the presence of polyubiquitin chains before NF-kB activation. Such structures may permit proximity-based trans-autophosphorylation, leading to cooperative activation of the signalling cascade. We further show that NF-kB activation results in modification of these structures. Finally, we demonstrate that these structures are abrogated in cells derived from incontinentia pigmenti patients.