Lien vers Pubmed [PMID] – 19247442
PLoS Pathog. 2009 Feb;5(2):e1000314
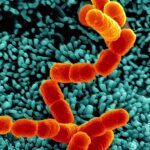
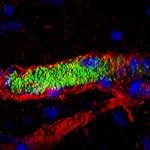
Bacterial infections targeting the bloodstream lead to a wide array of devastating diseases such as septic shock and meningitis. To study this crucial type of infection, its specific environment needs to be taken into account, in particular the mechanical forces generated by the blood flow. In a previous study using Neisseria meningitidis as a model, we observed that bacterial microcolonies forming on the endothelial cell surface in the vessel lumen are remarkably resistant to mechanical stress. The present study aims to identify the molecular basis of this resistance. N. meningitidis forms aggregates independently of host cells, yet we demonstrate here that cohesive forces involved in these bacterial aggregates are not sufficient to explain the stability of colonies on cell surfaces. Results imply that host cell attributes enhance microcolony cohesion. Microcolonies on the cell surface induce a cellular response consisting of numerous cellular protrusions similar to filopodia that come in close contact with all the bacteria in the microcolony. Consistent with a role of this cellular response, host cell lipid microdomain disruption simultaneously inhibited this response and rendered microcolonies sensitive to blood flow-generated drag forces. We then identified, by a genetic approach, the type IV pili component PilV as a triggering factor of plasma membrane reorganization, and consistently found that microcolonies formed by a pilV mutant are highly sensitive to shear stress. Our study shows that bacteria manipulate host cell functions to reorganize the host cell surface to form filopodia-like structures that enhance the cohesion of the microcolonies and therefore blood vessel colonization under the harsh conditions of the bloodstream.