Présentation
Computational implications of functional synaptic diversity – a formalized hypothesis.
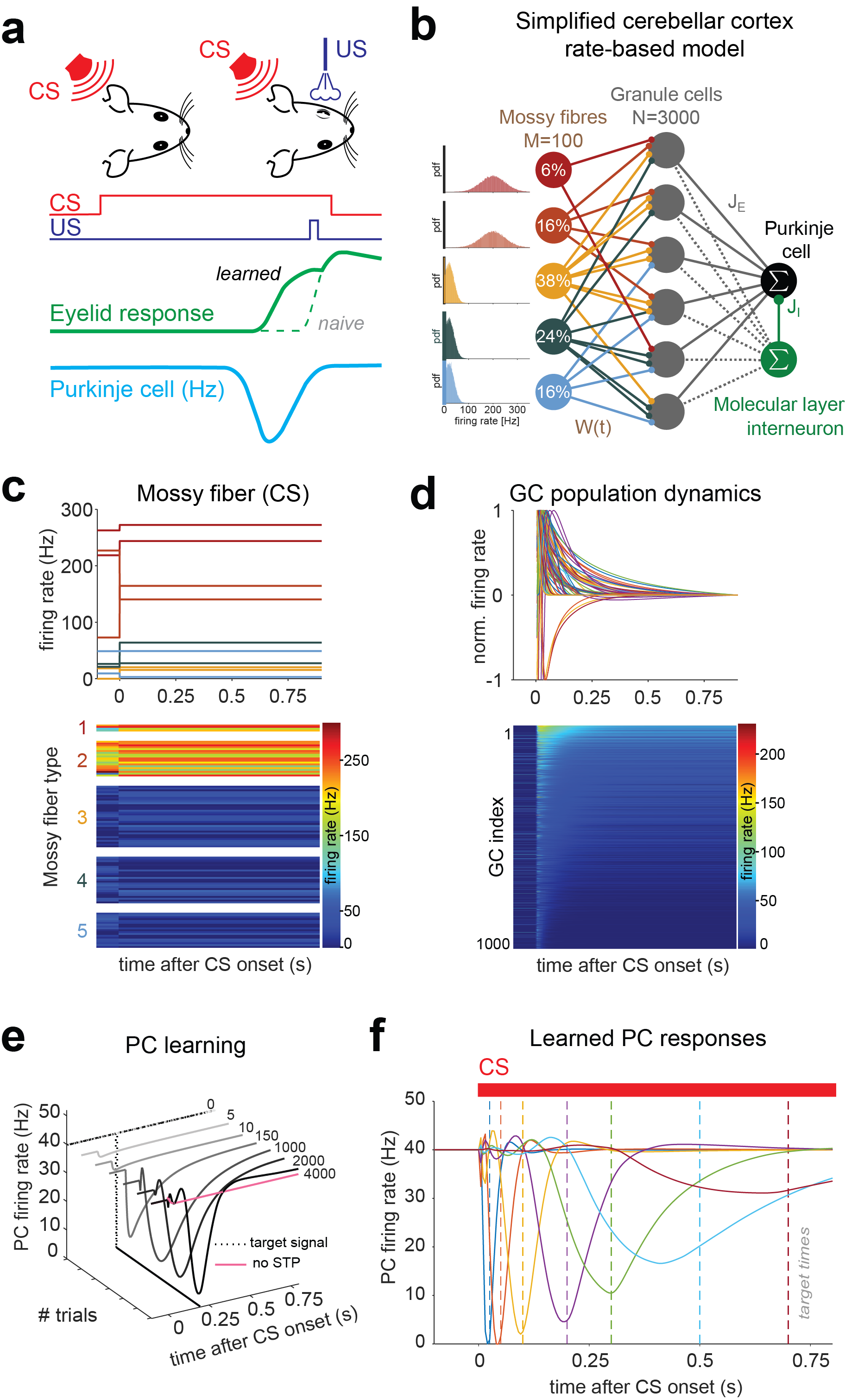
Precise neural representations of “time” are essential for well-timed behaviors [1], but the cellular and circuit mechanisms are largely unknown. We previously showed that the strength and short-term plasticity (STP) of synapses passing sensory information from mossy fibers (MFs) to granule cells (GCs) within the cerebellar cortex (CC) are highly variable, and can be a substrate for temporal coding of multimodal information. Since, we built a network model showing that MF-GC STP diversity generates heterogeneous GC spiking patterns, so-called temporal basis, which encodes elapsed time. The precise spiking output of the CC (Purkinje cells, PCs) can then be fine-tuned to drive precise behaviors by adjusting the strength (supervised learning) of those synapses from GCs with the relevant spiking dynamics (Figure 1). These model results therefore drive the hypothesis that dynamic synaptic diversity is necessary to encode elapsed time to perform precisely time behaviors.
Projects include incorporation of more accurate dynamic synapse models, a deeper examination of output performance with different synapse behaviors, and extension to include other circuit mechanisms such as molecular layer short-term plasticity.
Theoretical projects will examine the trade-off between temporal coding and pattern decorrelation.
Figure 1. Two-layer rate-based perceptron circuit model of the cerebellar cortex with dynamic synapses at the input layer.