Link to Pubmed [PMID] – 9234746
Infect. Immun. 1997 Aug;65(8):3003-10
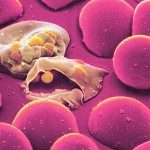
During Plasmodium falciparum asexual intraerythrocytic development, the host’s cell plasma membrane is modified by the insertion of parasite proteins. One or more of these modifications mediate the cytoadherence of infected erythrocytes to host vascular endothelium. However, these surface antigens can be the target of cytophilic antibodies which promote phagocytosis of the infected erythrocyte. It has been proposed that antibodies directed to epitopes rich in asparagine play an important role in this process, which has promoted efforts to isolate the corresponding gene(s). We describe here P. falciparum asparagine- and aspartate-rich protein 1 (PfAARP1), a new giant (circa 700-kDa) protein associated with the infected erythrocyte membrane which is rich in asparagine and aspartate residues due to the presence of nine blocks of repeats. Topology analysis predicts that PfAARP1 has multiple transmembrane domains and at least five external loops. Human antibodies immunopurified against a sequence composed exclusively of asparagine and aspartate amino acids derived from PfAARP1 label the surface of the infected erythrocyte, demonstrating that such motifs are exposed. Interestingly, external loop 4 of PfAARP1 contains repetitions of these residues, and their possible role as a target of cytophilic antibodies is discussed.